Which element has only one Subshell
On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1s orbital in their neutral, non-charged, state.
Which shell has only one subshell?
Subshell labelℓShells containing its0Every shellp12nd shell and higherd23rd shell and higherf34th shell and higher
Is a 1s subshell possible?
In the first shell, there is only the 1s orbital, as this shell can have a maximum of only 2 electrons. Therefore, the 1p orbital doesn’t exist. In the second shell, both 2s and 2p orbitals exist, as it can have a maximum of 8 electrons.
What subshell has 1 orbitals?
Specifies the orientation in space of an orbital of a given energy (n) and shape (l). This number divides the subshell into individual orbitals which hold the electrons; there are 2l+1 orbitals in each subshell. Thus the s subshell has only one orbital, the p subshell has three orbitals, and so on.How many Subshells does chlorine have?
SubshellMaximum Number of Electronsp6d10f14
How many Subshells are there in L shell?
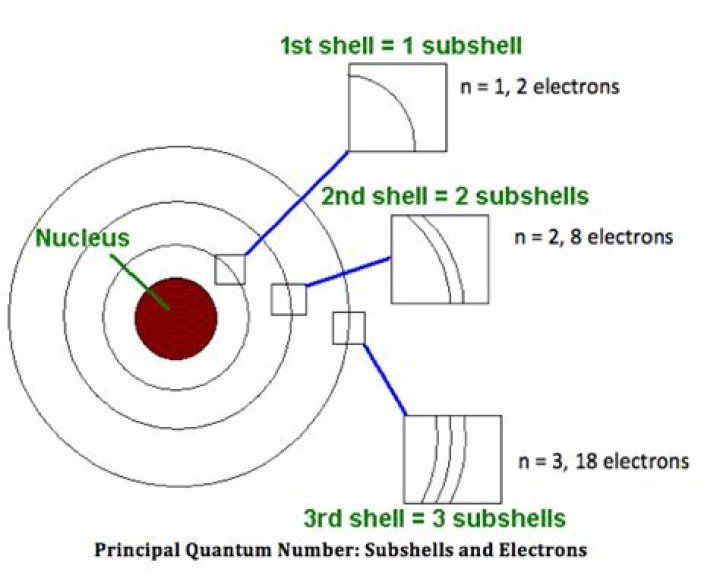
Each shell is composed of one or more subshells. The first K shell has one subshell, called ‘1s’; the L shell has two subshells, called ‘2s’ and ‘2p’; the third shell has ‘3s’, ‘3p’, and ‘3d’; and so on. A subshell is the set of states defined by azimuthal quantum number, l, within a shell.
What is a subshell in an atom?
A subshell is a subdivision of electron shells separated by electron orbitals. Subshells are labelled s, p, d, and f in an electron configuration.
Is Shell and Orbit same?
Orbit is a well-defined circular path around the nucleus in which electrons revolve around the nucleus. It is also called a shell. It is denoted by the principal quantum number ‘n’. Electrons can jump from one orbit to another by either absorbing energy or releasing energy.What is 1s 2s 2p 3s 3p?
In the question 1s 2s 2p 3s 3p represents electron orbital energy levels. … The sequence of orbital energy levels is as always-1s < 2s = 2p < 3s = 3p = 3d <4s = 4p = 4d= 4f. The orbital having the same energy is called a degenerate orbital.
How are atom shells filled?- Determine the number of electrons that the atom has.
- Fill the s orbital in the first energy level (the 1s orbital) with the first two electrons.
- Fill the s orbital in the second energy level (the 2s orbital) with the second two electrons.
Is 2p possible?
Among the given orbitals only 2s and 2p are possible. 1p and 3f cannot exist. For p-orbital, l = 1.
Why is 1p not possible?
The reason why there is no 1p orbital is that in the first shell only 1s is present because the shell can have maximum 2 electrons. Therefore, 1p is not possible.
Is 3p possible?
The 3p subshell contains three orbitals with 2 electrons each; therefore, there are six electrons. The principal quantum number (n) is 3, and the azimuthal quantum number (l) is 1. For the m quantum number, the possible values are n ± 1.
How do you write EC?
While writing electron configurations, a standardized notation is followed in which the energy level and the type of orbital are written first, followed by the number of electrons present in the orbital written in superscript. For example, the electronic configuration of carbon (atomic number: 6) is 1s22s22p2.
How many electrons does f have?
Fluorine is the ninth element with a total of 9 electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.
Where is the subshell?
SubshellNumber of OrbitalsMaximum Number of Electronsp36d510f714
How do you find the subshell?
Strategy. Subshells are determined by the value of l; thus, we first determine which values of l are allowed, and then we apply the equation “maximum number of electrons that can be in a subshell = 2(2l + 1)” to find the number of electrons in each subshell.
What is SPDF rule?
There are different orbital shapes (s,p,d,f) Each orbital can only hold 2 electrons max. There is a hierarchy, i.e. s orbitals will be filled before p orbitals which will be filled before d orbitals and so on. (s<p<d<f) (note, this is a general rule but there are exceptions)
What is the M shell?
Definition of M-shell : the third innermost shell of electrons surrounding an atomic nucleus — compare k-shell , l-shell.
Which Subshells are present in n shell?
They are represented as s, p, d, f. It is asked in the question to find out how many subshells are there in the N shell. So let us try to find out what might be the answer to this. subshells naming 4s, 4p, 4d and 4f.
Which subshell is filled after 4d?
The 4d sublevel is filled next, after the 5s sublevel.
How do you name Subshells?
ShellSubshellTotal Number of Electrons in Shell1st Shell1s22nd Shell2s, 2p2 + 6 = 83rd Shell3s, 3p, 3d2 + 6 + 10 = 18
What is the p sublevel?
The p sublevel has 3 orbitals, so can contain 6 electrons max. The d sublevel has 5 orbitals, so can contain 10 electrons max. And the 4 sublevel has 7 orbitals, so can contain 14 electrons max. … The p sublevels are called 2p, 3p, and 4p.
Who discovered Proton?
It is 100 years since Ernest Rutherford published his results proving the existence of the proton. For decades, the proton was considered an elementary particle.
What is a electron cloud?
Definition of electron cloud : the system of electrons surrounding the nucleus of an atom.
Are SPDF orbitals or subshells?
They are subshells. S-subshell only has one orbital – s-orbital.
Is orbital same as subshell?
A subshell is composed of orbitals. It is a subdivision of electron shells which is separated by electron orbitals. … Each subshell contains one or more orbitals. s contain one orbital, p contains 3 orbitals, d contains 5 orbitals and f contain 7 orbitals.
What are the D block elements called?
The d-block elements are found in groups 3, 4, 5, 6, 7, 8, 9, 10, 11, and 12 of the periodic table; d-block elements are also known as the transition metals. The d orbital is filled with the electronic shell “n-1.”
What is inside an electron?
Right now, our best evidence says that there are particles inside of neutrons and protons. Scientists call these particles quarks. Our best evidence also shows us that there is nothing inside of an electron except the electron itself.
Is 2s possible?
(ii) The second sub-shell has two subshells, i.e., 2s and 2p. Therefore, 2s orbitals are possible.
Can orbital 3f exist?
In the third shell, only the 3s, 3p and 3d orbitals exist, as it can hold a maximum of 18 electrons. Therefore, the 3f orbitals do not exist.