What makes a chair conformation stable
The chair conformation is more stable because it does not have any steric hindrance or steric repulsion between the hydrogen bonds. … Of these two positions of the H’s, the equitorial form will be the most stable because the hydrogen atoms, or perhaps the other substituents, will not be touching each other.
What makes a chair conformation less stable?
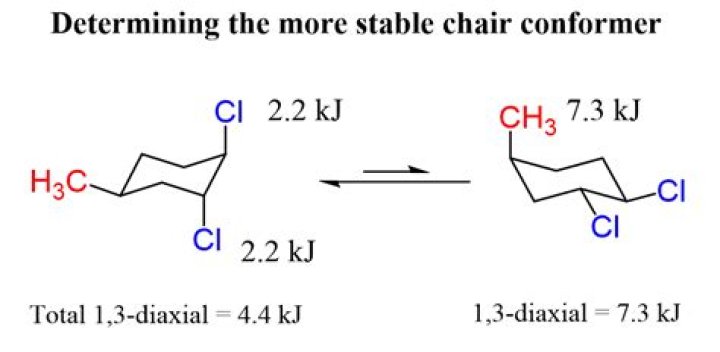
To Determine Chair Conformation Stability, Add Up The A-Values For Each Axial Substituent. The Lower The Number, The More Stable It is.
Why does cyclohexane adopt a chair conformation?
Explanation: The carbons in cyclohexane are all sp3 hybridized. The ideal angle between atoms connected to anything that is sp3 hybridized is 109.5 degrees. In a chair conformation, the angles of all the atoms in the ring can adopt this positioning, and thus the molecule does not experience any ring or angle strain.
Why is the chair conformation more stable than the boat?
Answer: Chair conformation of cyclohexane is more stable than boat form because in chair conformaion the C-H bonds are equally axial and equatorial, i.e., out of twelve C-H bonds, six are axial and six are equatorial and each carbon has one axial and one equatorial C-H bond.Is chair conformation a diastereomer?
The diequatorial chair conformer of the cis 1,3-dichloro isomer is achiral. … Both are achiral, since the disubstituted six-membered ring has a plane of symmetry. These isomers are diastereomers of each other, and are constitutional isomers of the 1,2- and 1,3- isomers.
Why is the half chair so unstable?
The half chair form is least stable due to maximum strain. It is 10kcal/mol less stable than the chair form and is least stable. The twist boat conformation has less steric and torsion strain as compared to half chair conformation and It is 5.5kcal/mol less stable than the chair form.
How do you make cyclohexane?
Modern. On an industrial scale, cyclohexane is produced by hydrogenation of benzene in the presence of a Raney nickel catalyst. Producers of cyclohexane account for approximately 11.4% of global demand for benzene. The reaction is highly exothermic, with ΔH(500 K) = -216.37 kJ/mol).
Why is gauche conformation less stable?
Answer: c Explanation: Gauche conformation is less stable due to Vander Waal’s repulsion. Vander Waal’s are weak forces of attraction which results in lesser stability of gauche conformer.Why are half chairs less stable than boats?
ii. The half chair, formed by raising the footrest of the chair, has five of the six C atoms in a plane and one C atom out of the plane. Therefore, it has both eclipsing and bond angle strains and hence is the least stable conformation of cyclohexane.
Is Half chair more stable than boat?The chair conformation is more stable than the boat conformation. The boat conformation can sometimes be more stable than it is usually, by a slight rotation in the C-C bonds and is called the skew boat conformation. Nevertheless, the chair conformation is the most stable cyclohexane form.
Article first time published onWhy is cyclohexane more stable than hexane?
This is due to the fact that cyclohexane has 2 hydrogens less compared to hexane. The volume cyclohexane occupies is lower than hexane due to its cyclic structure. Hence, there’s a higher number of cyclohexane molecules in a given molecule when compared with hexane.
Which of the conformation of cyclohexane is more stable?
The chair conformation is the most stable conformer. At 25 °C, 99.99% of all molecules in a cyclohexane solution adopt this conformation.
Is cis Equatorial?
Because of the alternating nature of equatorial and axial bonds, the opposite relationship is true for 1,3-disubstitution (cis is all equatorial, trans is equatorial/axial).
How can you tell if a chair conformation is meso?
Meso compounds are characterized by an internal plane of symmetry that renders them achiral despite the presence of chiral center(s). For the given six member rings, the key to identifying the meso compound is finding the structure in which the two chlorine atoms are on the same side of the ring.
How do you number carbons in a chair conformation?
Number the carbons in your cyclohexane and in your chair. Clockwise or counterclockwise doesn’t matter, as long as you use the same direction for both molecules. Then simply compare. Identify the carbon number for the first substituent, if it’s wedged add it to the up position.
What is the relation between hexene and cyclohexane?
Hexene and Cyclohexane have no relation between them. They are unrelated. Hexene and Cyclohexane are not isobars because isobars are the atoms having different masses but same atomic number. The atomic number of hexene is C6H12 and the atomic number of cyclohexane is C6H10.
How many equatorial hydrogen does a chair conformation carry?
On careful examination of a chair conformation of cyclohexane, we find that the twelve hydrogens are not structurally equivalent. Six of them are located about the periphery of the carbon ring, and are termed equatorial.
Which chair conformation is lowest in energy?
The lower energy chair conformation is the one with three of the five substituents (including the bulky –CH2OH group) in the equatorial position. Exercise 3.7: Draw the two chair conformations of the six-carbon sugar mannose, being sure to clearly show each non-hydrogen substituent as axial or equatorial.
Which of the following chair conformations is the most stable?
The most stable conformation of cyclohexane is the chair form shown to the right. The C-C-C bonds are very close to 109.5o, so it is almost free of angle strain. It is also a fully staggered conformation and so is free of torsional strain.
What is the order of stability of confirmation?
Fully eclipsed>staggered>partially eclipsed>gauche.
Is twist boat more stable than half chair?
The twist relieves some of the torsional strain of the boat and moves the flagpole H further apart reducing the steric strain. Consequently the twist boat is slightly more stable than the boat.
Which of the following conformation has highest stability?
hydrocarbons. …with respect to the other—the eclipsed conformation is the least stable, and the staggered conformation is the most stable. The eclipsed conformation is said to suffer torsional strain because of repulsive forces between electron pairs in the C―H bonds of adjacent carbons.
Is axial or equatorial more stable?
A conformation in which both substituents are equatorial will always be more stable than a conformation with both groups axial.
Which is a chair conformation of cyclohexane?
The most stable conformation of cyclohexane is called “chair“ conformation, since it somewhat resembles a chair. In the chair conformation of cyclohexane, all the carbons are at 109.5º bond angles, so no angle strain applies.
Can gauche be more stable than anti?
These conformations differ by the relative positions of the two methyl substituents. … This is called the gauche conformation. The gauche form is less stable than the anti form due to steric hindrance between the two methyl groups but still is more stable than the eclipsed formations.
Why staggered is more stable?
From the structures of staggered and eclipsed conformations, we can conclude that the torsional strain in the staggered conformation is less than that of the eclipsed conformation. Thus, the staggered conformation is more stable than the eclipsed conformation because staggered conformation has no torsional strain.
Why is cyclopentane unstable?
The smaller the carbon ring the more unstable it is. The reason for this is that the molecule cannot freely rotate about each bond. … This molecule has the highest ring strain of all the carbon rings. A five membered ring such as cyclopentyne is not too far from that.
Which is more stable Cycloheptane or cyclopentane?
Cyclopentane is slightly more stable because it can relieve some of the bond strain by buckling the ring and pushing one carbon out of the plane.
Are chains or rings more stable?
It is found that for the nitrogen-rich molecules, the structures with N3-chains (N5-ring) are more stable than those containing a N3-ring (N5-chain). … Energy decomposition analysis shows the bond of boron−nitrogen is stronger than that of carbon−nitrogen.
Which is more stable cyclohexane or cyclopentane?
Cyclopentane is a more stable molecule with a small amount of ring strain, while cyclohexane is able to adopt the perfect geometry of a cycloalkane in which all angles are the ideal 109.5° and no hydrogens are eclipsed; it has no ring strain at all.
Which of a )-( D is the most stable conformation?
So, the most stable conformation of cyclohexane is the chair conformation (Option D).