What is true pepsin
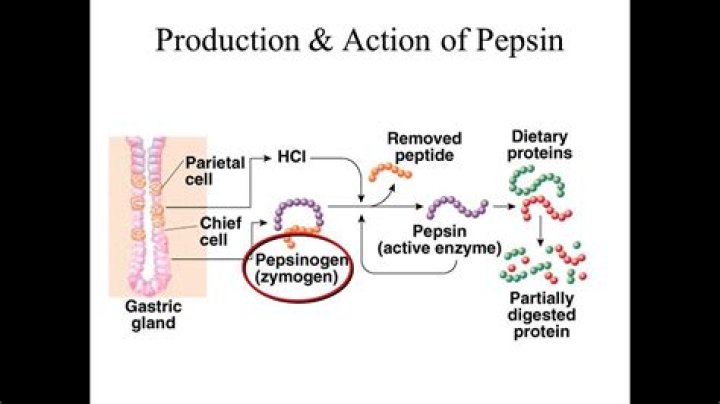
Pepsin is a stomach enzyme that serves to digest proteins found in ingested food. Gastric chief cells secrete pepsin as an inactive zymogen called pepsinogen. Parietal cells within the stomach lining secrete hydrochloric acid that lowers the pH of the stomach. A low pH (1.5 to 2) activates pepsin.
What's true about pepsin?
Pepsin is an endopeptidase that breaks down proteins into smaller peptides. It is produced in the gastric chief cells of the stomach lining and is one of the main digestive enzymes in the digestive systems of humans and many other animals, where it helps digest the proteins in food.
What is pepsin short answer?
An enzyme made in the stomach that breaks down proteins in food during digestion. Stomach acid changes a protein called pepsinogen into pepsin.
Which is true concerning the enzyme pepsin?
Correct answer: Pepsin is an enzyme in the stomach that digests proteins. Because it is active in the stomach, which is highly acidic, pepsin best functions at a low pH between 2 and 2.5. Pepsinogen is secreted by chief cells and converted into active pepsin after catalyzation by hydrochloric acid.What is an example of a pepsin?
pepsin, the powerful enzyme in gastric juice that digests proteins such as those in meat, eggs, seeds, or dairy products. Pepsin is the mature active form of the zymogen (inactive protein) pepsinogen. Pepsin was first recognized in 1836 by the German physiologist Theodor Schwann.
What is the main role of pepsin in this metabolic reaction?
Pepsin is an enzyme that acts in the stomach for protein digestion. Which of these is most likely the main role of pepsin in this metabolic reaction? Enzymes enable reactions to occur at lower temperatures.
What does pepsin do to proteins?
Of these five components, pepsin is the principal enzyme involved in protein digestion. It breaks down proteins into smaller peptides and amino acids that can be easily absorbed in the small intestine.
What enzyme helps digest eggs?
pepsin — breaks down proteins in foods like meat, eggs, and dairy products into smaller peptides.What are the functions of pepsin?
The Function Of Pepsin are: It helps in the digestion. It breaks down the large polypeptides into smaller peptide fragments. Help break down dietary proteins like eggs, meat, seeds, and other dairy products.
Why pepsin works best at pH 2?The reason pepsin functions best at pH 2 is because the carboxylic acid group on the amino acid in the enzyme’s active site must be in its protonated state, meaning bound to a hydrogen atom. At low pH the carboxylic acid group is protonated, which allows it to catalyze the chemical reaction of breaking chemical bonds.
Article first time published onWhat is pepsin substrate?
The substrate of the enzyme pepsin is protein. Pepsin is actually an aspartic protease as aspartate is present in the active site for the catalysis…
Is pepsin intracellular or extracellular?
Question : Pepsin is an intracellular/extracellular enzyme. Which is correct? Video Solution: Pepsin is an intracellular/extracellular enzyme.
Where can I find pepsin?
Pepsin can be found in gastric juices that are acidic and needed to properly metabolize the foods we eat. Glands in the mucous-membrane lining of the stomach, called peptic chief cells, are responsible for making pepsinogen.
Where is aminopeptidase active?
Aminopeptidases catalyze the cleavage of amino acids from the amino terminus of protein or peptide substrates. They are widely distributed throughout the animal and plant kingdoms and are found in many subcellular organelles, in cytoplasm, and as membrane components.
Does pepsin do hydrolysis?
Pepsin is an enzyme belonging to the family of aspartic protease enzymes. … The specific reaction catalyzed by pepsin is the acid hydrolysis of the peptide bond. This reaction will break down proteins into smaller units to enable the digestive process.
Are peptides and pepsin the same?
is that peptide is (biochemistry) a class of organic compounds consisting of various numbers of amino acids in which the amine of one is reacted with the carboxylic acid of the next to form an amide bond while pepsin is (enzyme) a digestive enzyme that chemically digests, or breaks down, proteins into shorter chains of …
Does pepsin produce amino acids?
The activation of pepsin is accomplished by hydrolysis of the peptide bond between residues 42 and 43 of the zymogen, releasing a 42 amino acid segment from the N-terminus of the protein. Active pepsin has a mass of 35 kDa.
How many amino acids are in pepsin?
One subunit of 326 amino acids comprises pepsin, which has a molecular weight of 34,509.83g and an isoelectric point of 3.24(3). Pepsin is classified as bilobal, consisting of two domains which become close in proximity upon proper protein folding.
Does pepsin digest carbohydrates?
Protein digestion occurs in the stomach and the duodenum through the action of three main enzymes: pepsin, secreted by the stomach, and trypsin and chymotrypsin, secreted by the pancreas. During carbohydrate digestion the bonds between glucose molecules are broken by salivary and pancreatic amylase.
How protein gets metabolized?
Protein metabolism occurs in liver, specifically, the deamination of amino acids, urea formation for removal of ammonia, plasma protein synthesis, and in the interconversions between amino acids.
Where is protein processed in the body?
The bottom line Protein is a vital nutrient for almost every part of your body. It’s digested in your mouth, stomach, and small intestine before it’s released into your bloodstream as individual amino acids.
How are amino acids metabolized?
The catabolism of amino acids, except those with branched chains, starts in the liver. The amine group is separated and incorporated into urea. The carbon skeletons can be oxidized to CO2 and H2O or used for gluconeogenesis and ketogenesis. The liver is very efficient in the removal of ammonia.
What enzymes are produced in the stomach?
Region of digestive systemEnzymeWhere producedStomachProtease – pepsinGastric glands in stomachSmall intestine – DuodenumProtease – trypsinPancreasSmall intestine – IleumProtease – peptidaseWall of ileum
Is Potato hard to digest?
It’s also important to remove the potato skin because this can be hard to digest, says Carrie Dennett, MPH, a registered dietitian nutritionist and owner of Nutrition By Carrie.
Are avocados hard to digest?
Avocados: Though a rich source of dietary fiber, avocados should be avoided if you have an upset stomach. A part of the plant foods passes through your system without being digested.
Is cheese hard to digest?
Dairy products If you’re lactose intolerant, dairy may upset your digestion or cause diarrhea. Look for products that are lactose-free or low in lactose. Otherwise, dairy is low in fiber and may be easy to digest for many people. Try drinking plain milk or snacking on cheese, yogurt, and cottage cheese.
How does pH affect pepsin?
Pepsin is an aspartic protease and its activity is directly dependent on the pH of the solution environment [41]. At pH values >2.5, pepsin activity begins to decline, with incomplete degradation of the three susceptible substrate proteins by pepsin being observed.
What would be the optimum pH for pepsin?
Pepsin is an aspartic protease that acts in food digestion in the mammal stomach. An optimal pH of around 2 allows pepsin to operate in its natural acidic environment, while at neutral pH the protein is denatured.
What is the normal pH of gastric fluid?
The normal volume of the stomach fluid is 20 to 100 mL and the pH is acidic (1.5 to 3.5). These numbers are converted to actual acid production in units of milliequivalents per hour (mEq/hr) in some cases.
What are the 4 main digestive enzymes?
- Amylase.
- Maltase.
- Lactase.
- Lipase.
- Proteases.
- Sucrase.
Where does pepsin cleave?
Pepsin cleaves peptide bonds in the amino-terminal side of the cyclic amino acid residues (tyrosine, phenylalanine, and tryptophan), breaking the polypeptide chains into smaller peptides (Fange and Grove, 1979).