What is true about the subatomic particles
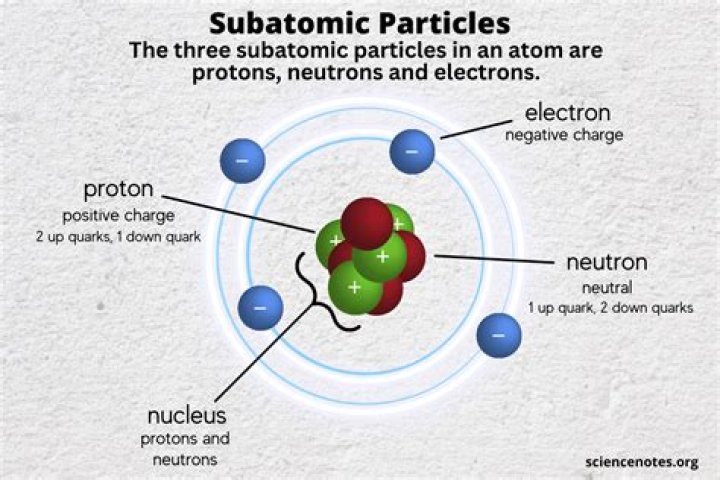
Subatomic particles include electrons, the negatively charged, almost massless particles that nevertheless account for most of the size of the atom, and they include the heavier building blocks of the small but very dense nucleus of the atom
What is true about the masses of subatomic particles?
Masses for the three subatomic particles can be expressed in amu (atomic mass units) or grams. For simplicity, we will use the amu unit for the three subatomics. Both neutrons and protons are assigned as having masses of 1 amu each. In contrast, the electron has a negligible mass of .
Which of the following is not true about subatomic particles in an atom?
The statement that is not true about subatomic particle is that all electrons and atom contain the same amount of energy.
What makes up a subatomic particle?
These particles include atomic constituents such as electrons, protons, and neutrons (protons and neutrons are actually composite particles, made up of quarks), as well as other particles such as photons and neutrinos which are produced copiously in the sun. …Which statement is true about subatomic particles is true?
The true statement about subatomic particles is A) A neutral atom contains the same number of protons and electrons.
What are the function roles of the subatomic particles in an atom?
The main three subatomic particles are Protons, electrons and neutrons. … Protons, including neutrons, give the atom mass but are not involved in chemical reactions. The number of protons that an atom has is called the atomic number and determines the identity of the atom.
Which of the following is true about subatomic particles quizlet?
Which of the following is true about subatomic particles? Electrons are negatively charged and are the heaviest subatomic particle. Protons are positively charged and the lightest subatomic particle. Neutrons have no charge and are the lightest subatomic particle.
Which statements about subatomic particles are false?
Protons and neutrons have charges of the same magnitude, but opposite sign. This is also false because neutrons have no charge, so it’s impossible for protons and neutrons to have the same charge.What are the subatomic particles in the nucleus?
The nucleus itself is generally made of protons and neutrons but even these are composite objects. Inside the protons and neutrons, we find the quarks, but these appear to be indivisible, just like the electrons.
Which of the following is not a subatomic particle?Thus, we can say that there are three sub- atomic particles namely electrons, protons and neutrons. Nucleus is not a sub- atomic particle.
Article first time published onWhich of the following is a subatomic particle?
Protons, neutrons, and electrons are the three main subatomic particles found in an atom.
Which statement about subatomic particles is true protons neutrons and electrons all have about the same mass?
Protons, neutrons, and electrons all have about the same mass. Unlike protons or neutrons, electrons have no mass. Neutrons have no charge and no mass. An electron has far less mass than either a proton or neutron.
Which statement best describes the location of subatomic particles?
Which statement best describes the location of subatomic particles? The protons and electrons are in the nucleus, while the neutrons are in the cloud outside the nucleus.
Which of the following are true about protons?
ParticleSymbolLocationprotonp+inside the nucleuselectrone−outside the nucleusneutronn0inside the nucleus
What did Thomson believe about atoms?
Summary. J.J. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.”
Are subatomic particles bigger than atoms?
In physical sciences, a subatomic particle is a particle that is smaller than an atom.
Which subatomic particle occupies almost all of an atom's volume?
The nucleus contains the majority of an atom’s mass because protons and neutrons are much heavier than electrons, whereas electrons occupy almost all of an atom’s volume.
What is the significance of the three subatomic particles?
Protons, neutrons and electrons make up the subatomic particles of an atom. The three subatomic particles determine the overall charge of an atom, the chemical characteristics it can possess and its physical properties.
What is the most important subatomic particle and why?
I would say proton and electron are the two most important subatomic particles in an atom. The number of protons inside a nucleus must equal to the number of electrons outside of the nucleus. This number determines all the chemical properties of the atom, which governs the behaviors of all matters in our world.
How do the subatomic particles affect the properties of an atom?
A neutral atom has an equal number of protons and electrons. Because electrons orbit around atomic nuclei, they are the subatomic particles that affect chemical reactions. Loss of electrons can lead to the formation of positive-charged species called cations. Gaining electrons can yield negative species called anions.
What is the mass of subatomic particles?
ParticleElectric Charge (C)Mass (g)Protons+1.6022 x 10-191.6726 x 10-24Neutrons01.6740 x 10-24Electrons-1.6022 x 10-199.1094 x 10-28
Can you see subatomic particles?
We can never see the subatomic particles directly, but can only infer from observation of such indirect effects like tracks. If there are many of them and they are emitting some radiation, and also if we shine some radiation on then and receive back the response this will also constitute a kind of seeing.
What order were subatomic particles?
The first subatomic particle to be discovered was the electron, identified in 1897 by J. J. Thomson. After the nucleus of the atom was discovered in 1911 by Ernest Rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. In 1932 the neutron was discovered.
What are three subatomic particles of an atom?
Given that these particles make up atoms, they are often referred to as subatomic particles. There are three subatomic particles: protons, neutrons and electrons. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge.
Which statement about subatomic particles are true select all that apply?
Protons are positively charged. Thomson’s experiments with cathode rays determined that atoms contained even smaller particles called _____, which have a _____ charge. Which of the following statements correctly describe Rutherford’s experiment? Select all that apply.
What is proton and neutron?
Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).
What is AMU based on?
An atomic mass unit (symbolized AMU or amu) is defined as precisely 1/12 the mass of an atom of carbon-12. The carbon-12 (C-12) atom has six protons and six neutrons in its nucleus. In imprecise terms, one AMU is the average of the proton rest mass and the neutron rest mass.
Is molecule a subatomic particle?
A subatomic particle is a particle smaller than an atom. This means it is very, very small. Like atoms and molecules, a subatomic particle is far too small to be seen with the naked eye. … The commonly studied subatomic particles are the main ones that make atoms: protons, neutrons, and electrons.
Which of the following is not a subatomic particle found in the nucleus of atoms?
The correct option is c. The nucleus only contains protons and neutrons. It does not contain electrons. Electrons surround the nucleus and are present in the atomic orbitals.
Is photon a subatomic particle?
Considered among the subatomic particles, photons are bosons, having no electric charge or rest mass and one unit of spin; they are field particles that are thought to be the carriers of the electromagnetic field.
What are the subatomic particles quizlet?
The three subatomic particles are protons, neutrons, and electrons. … The nucleus contains protons and neutrons.