What is the role of tetrahydrobiopterin
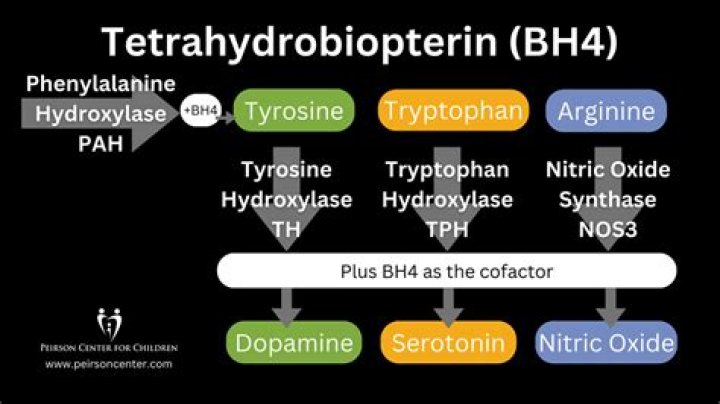
Tetrahydrobiopterin (BH4, THB), also known as sapropterin (INN), is a cofactor of the three aromatic amino acid hydroxylase enzymes, used in the degradation of amino acid phenylalanine and in the biosynthesis of the neurotransmitters serotonin (5-hydroxytryptamine, 5-HT), melatonin, dopamine, norepinephrine ( …
What enzyme requires tetrahydrobiopterin as a cofactor?
Tetrahydrobiopterin is an essential cofactor for the neurotransmitter synthesizing enzymes tyrosine hydroxylase (which catalyzes the conversion of tyrosine to l-dopa) and tryptophan hydroxylase (which catalyzes the conversion of tryptophan to 5-hydroxytryptophan [5-HTP]), as well as for phenylalanine hydroxylase (which …
Where is BH4 from?
BH4 is made from the molecule GTP (guanosine triphosphate). GTP is converted into BH4 in three stages, which are catalysed (in order) by the enzymes GTPCH, PTPS and SR. These enzymes are coded for, respectively, by the GCH1, PTS and SR genes. Interestingly, rare mutations in these genes can lead to deficiency of BH4.
What is the Biopterin pathway?
Synthesis. Biopterin synthesis occurs through two principal pathways; the de novo pathway involves three enzymatic steps and proceeds from GTP, while the salvage pathway converts sepiapterin to biopterin. BH4 is the principal active cofactor, and a recycling pathway converts BH2 to BH4.How is BH4 formed?
BH4 is formed de novo from GTP via a sequence of three enzymatic steps carried out by GTP cyclohydrolase I, 6-pyruvoyltetrahydropterin synthase and sepiapterin reductase. An alternative or salvage pathway involves dihydrofolate reductase and may play an essential role in peripheral tissues.
How Tetrahydrobiopterin is essential for the synthesis of CNS neurotransmitters?
Tetrahydrobiopterin is an essential cofactor for the neurotransmitter synthesizing enzymes tyrosine hydroxylase (which catalyzes the conversion of tyrosine to l-dopa) and tryptophan hydroxylase (which catalyzes the conversion of tryptophan to 5-hydroxytryptophan [5-HTP]), as well as for phenylalanine hydroxylase (which …
What is the role of tetrahydrobiopterin in the synthesis of serotonin?
Tetrahydrobiopterin has multiple roles in human biochemistry. The major one is to convert amino acids such as phenylalanine, tyrosine, and tryptophan to precursors of dopamine and serotonin, major monoamine neurotransmitters.
What does Hyperphenylalaninemia mean?
Hyperphenylalaninemia is broadly defined as the presence of blood phenylalanine levels that exceed the limits of the upper reference range (2 mg/dL or 120 µmol/L) without treatment but that are below the level found in patients with phenylketonuria (PKU).What is the importance of Biopterin?
Biopterin is an important co-enzyme for breaking down amino acids, especially the amino acid phenylalanine. BIOPT-BS occurs when the body cannot make its own BH4, the substance from which biopterin is made. Babies with BIOPT-BS either do not produce enough or produce non-working enzymes for making BH4.
What is neonatal Tyrosinemia?Transient tyrosinemia of the newborn is a benign disorder of tyrosine metabolism detected upon newborn screening and often observed in premature infants. It shows no clinical symptoms. It is characterized by tyrosinemia, moderate hyperphenylalaninemia, and tyrosiluria that usually resolve after 2 months of age.
Article first time published onHow does tyrosine work?
Supplementing with tyrosine is thought to increase levels of the neurotransmitters dopamine, adrenaline and norepinephrine. By increasing these neurotransmitters, it may help improve memory and performance in stressful situations (4). Summary Tyrosine is an amino acid that the body produces from phenylalanine.
How is BH4 deficiency treated?
Treatment of BH4 deficiencies consists of BH4 supplementation (2-20 mg/kg per day) or diet to control blood phenylalanine concentration and replacement therapy with neurotransmitters precausers (L-dopa/CarbiDOPA and 5-hydroxytryptophan), and supplements of folinic acid in DHPR deficiency.
What enzyme converts dopa to dopamine?
DOPA is converted to dopamine by aromatic amino acid decarboxylase. Dopamine-β-hydroxylase hydroxylates dopamine to norepinephrine, which is methylated to epinephrine by phenylethanolamine N-methyltransferase. Tyrosine hydroxylase is the rate-limiting enzyme of the pathway.
What is the function of the BH4 gene?
The most well-established human function of tetrahydrobiopterin (BH4) is as the cofactor for phenylalanine-4-hydroxylase (PAH), tyrosine-3-hydroxylase, and tryptophan-5-hydroxylase. The latter two are key enzymes in biogenic amine biosynthesis (ie, aromatic amino acid synthesis).
What is the shape of BH4?
The shape of BH−4 is tetrahedral because in BH−4 , boron is sp3 hybridised.
What is the overall charge of BH4?
Example: the ion BH4– has an overall charge of -1. This means that the formal charges of all the individual atoms in it should add up to -1. Let’s see if this is true. Boron has three valence electrons, eight bonded electrons, and zero unbonded electrons.
Why is phenylalanine converted to tyrosine?
To enter the tyrosine pathway, phenylalanine is converted into tyrosine by the enzyme phenylalanine hydroxylase, which has tetrahydrobiopterin as a cofactor. Deficiency of the enzyme or of its cofactor causes accumulation of phenylalanine in the body fluids and tissues.
What is phenylalanine made from?
Good sources of phenylalanine are eggs, chicken, liver, beef, milk, and soybeans. Another common source of phenylalanine is anything sweetened with the artificial sweetener aspartame, such as diet drinks, diet foods and medication; the metabolism of aspartame produces phenylalanine as one of the compound’s metabolites.
What is H4 Biopterin?
Tetrahydrobiopterin (H4-biopterin) is an essential cofactor of a set of enzymes that are of central metabolic importance, i.e. the hydroxylases of the three aromatic amino acids phenylalanine, tyrosine, and tryptophan, of ether lipid oxidase, and of the three nitric oxide synthase (NOS) isoenzymes.
Is tyrosine an enzyme?
Tyrosine ammonia lyase (TAL) is an enzyme in the natural phenols biosynthesis pathway.
Is Hyperphenylalaninemia the same as PKU?
Benign hyperphenylalaninemia (H-PHE) is a mild form of phenylketonuria. It is considered an amino acid condition because people with H-PHE have problems breaking down an amino acid, a building block of proteins, known as phenylalanine. Most people with this condition experience mild symptoms or no symptoms.
What is the difference between PKU and Hyperphenylalaninemia?
HyperphenylalaninemiaSpecialtyEndocrinology
What is normal level of phenylalanine?
Normal levels of phenylalanine in the blood are less than 2 milligrams per deciliter (mg/dL). More than 4 mg/dL of phenylalanine in the blood is considered high and may mean your child has PKU.
What is the treatment for tyrosinemia?
The treatment for tyrosinemia is a combination of a low-protein diet and a drug called Nitisinone. Nitisinone prevents the build up of toxic breakdown products. Meats, dairy products, and other protein rich foods such as nuts and beans should be avoided.
How is tyrosinemia caused?
Tyrosinemia is caused by mutations in the fumarylacetoacetate hydrolase (FAH) gene that is responsible for the production of the FAH enzyme.
What are the three types of tyrosinemia?
Mutations in the FAH, TAT, and HPD genes can cause tyrosinemia types I, II, and III, respectively. In the liver , enzymes break down tyrosine in a five step process, resulting in molecules that are either excreted by the kidneys or used to produce energy or make other substances in the body.
Does tyrosine help thyroid?
Supplementation with L-tyrosine (one of its naturally occurring isomers) is commonly used to support thyroid function. Given its role in thyroxin production, tyrosine availability could theoretically affect thyroid function.
Does tyrosine increase testosterone?
L-tyrosine also enhances testosterone levels and semen quality of bulls (16).
Does tyrosine increase heart rate?
Small doses of tyrosine produce tachycardia and hypertension while higher doses produce bradycardia and hypotension in anaesthetised rats. The mechanism of these effects has not been established. An increased synthesis and release of catecholamines has been suggested to be the mechanism.
What is tetrahydrobiopterin deficiency?
Tetrahydrobiopterin deficiency is a rare disorder characterized by a shortage (deficiency) of a molecule called tetrahydrobiopterin or BH4. This condition alters the levels of several substances in the body, including phenylalanine.
How can PKU be prevented?
- Follow a low-phenylalanine diet. Women with PKU can prevent birth defects by sticking to or returning to a low-phenylalanine diet before becoming pregnant. …
- Consider genetic counseling.