What is the product of hydrogenation
The most commonly hydrogenated product is vegetable oil. Hydrogenation converts vegetable oil from a liquid to a solid or semi-solid fat. D-Sorbitol syrup is made by hydrolysis of starches to make dextrose, the dextrose is then hydrogenated to create sorbitol or sugar alcohol.
What is the end product of hydrogenation called?
At high temperatures, during hydrogenation, the double bond can rearrange (or change positions) in unsaturated hydrocarbons. If they rearrange, the end result may be a product that still contains double bonds. In nutrition we call these products, following hydrogenation, trans fats.
What compounds are produced by hydrogenation?
Organic chemistry Hydrogenation is a useful means for converting unsaturated compounds into saturated derivatives. Substrates include not only alkenes and alkynes, but also aldehydes, imines, and nitriles, which are converted into the corresponding saturated compounds, i.e. alcohols and amines.
What is the product of hydrogenation of an alkene?
The product is an alkane. Hydrogenation is used in the food industry to convert liquid oils into saturated fats. This process yields semi-solid products like shortening and margarine.What is hydrogenation give an example?
Hydrogenation is a chemical reaction that adds hydrogen to a molecule. Hydrogenation is not thermodynamically favorable at ordinary temperatures, so a catalyst is needed. Usually this catalyst is a metal. Examples of hydrogenated products include margarine, mineral turpentine, and aniline.
What does hydrogenation mean?
Hydrogenation is a chemical reaction between molecular hydrogen and an element or compound, ordinarily in the presence of a catalyst. … The catalysts most commonly used for hydrogenation reactions are the metals nickel, platinum and palladium and their oxides.
What is hydrogenation alkane?
In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane. Hydrogenation of a double bond is a thermodynamically favorable reaction because it forms a more stable (lower energy) product.
What is the mechanism for hydrogenation?
In chemistry hydrocyanation is a process for conversion of alkenes to nitriles. The reaction involves the addition of hydrogen cyanide and requires a catalyst. This conversion is conducted on an industrial scale for the production of precursors to nylon.What is the product produced from the halogenation of alkenes?
The halides add to neighboring carbons from opposite faces of the molecule. The resulting product is a vicinal (neighboring) dihalide.
What is the product of hydrogenation of 1/2 Dimethylcyclopentene?What is(are) the product(s) in the Pd-catalyzed hydrogenation if 1,2-dimethylcyclopentene? Explanation: The product for this hydrogenation is cis-1,2-dimethylcyclopentane. It is important to note the three main types of reactions for alkenes.
Article first time published onHow are hydrogenated fats made?
Most trans fat is formed through an industrial process that adds hydrogen to vegetable oil, which causes the oil to become solid at room temperature. This partially hydrogenated oil is less likely to spoil, so foods made with it have a longer shelf life.
What is produced by hydrogenating unsaturated fat quizlet?
Hydrogenation of unsaturated fats produces some trans-fatty acids, so they are found most often in commercially fried foods and in baked goods and other commercial foods made with partially hydrogenated vegetable oils.
What is needed for hydrogenation?
Hydrogenation reactions generally require three components: the substrate, the hydrogen source, and a catalyst. The reaction is carried out at varying temperatures and pressures depending on the catalyst and substrate used. The hydrogenation of an alkene produces an alkane.
What is hydrogenation BYJU's?
Answer: Hydrogenation is an addition reaction between the hydrogen and other compounds in the presence of a catalyst. Example: Hydrogenation of ethene involves the addition of two hydrogen atoms across the double bond of ethene as a result saturated ethane is formed.
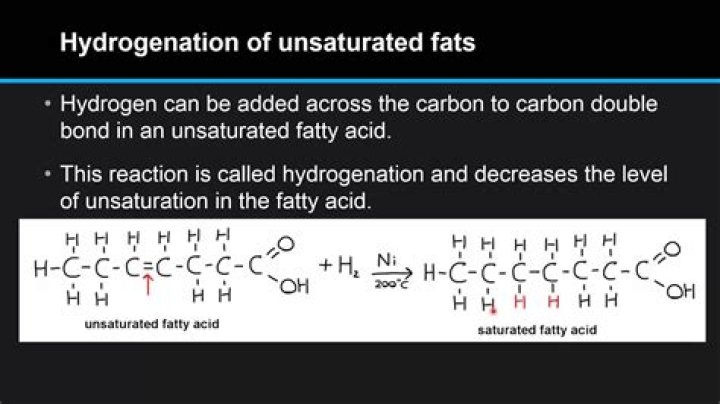
What is the product of the reaction between an unsaturated fatty acid and hydrogen in the presence of a metal catalyst?
A major health concern during the hydrogenation process is the production of trans fats. Trans fats are the result of a side reaction with the catalyst of the hydrogenation process. This is the result of an unsaturated fat which is normally found as a cis isomer converts to a trans isomer of the unsaturated fat.
Which substance is produced by the reaction of hydrogen with a vegetable oil?
It includes the manufacture of margarine from animal or vegetable fats and oils. Ethene reacts with hydrogen in the presence of a finely divided nickel catalyst at a temperature of about 150°C. Ethane is produced.
What is hydrogenation of benzene?
Hydrogenation is an addition reaction in which hydrogen atoms are added all the way around the benzene ring. … With benzene: . . . and methylbenzene: These reactions destroy the electron delocalisation in the original benzene ring, because those electrons are being used to form bonds with the new hydrogen atoms.
What will be the product formed by the complete hydrogenation of alkynes?
When an alkyne is hydrogenated, we are simply adding hydrogen atoms across the triple bond so that either a double bond remains, or only single bonds are present in the compound. If the final product contains a carbon-carbon double bond, it is called an alkene. If all single bonds are present, an alkane is produced.
What products is obtained when acetylene undergoes hydrogenation in presence of Pd Pt Ni?
Answer: Ammonia, fuels (hydrocarbons), alcohols, pharmaceuticals, margarine, polyols, various polymers and chemicals (hydrogen chloride and hydrogen peroxide) are products treated using a hydrogenation process.
What are the benefits of hydrogenation?
The hydrogenation process increases the melting point of the fat, which changes liquid oil into solid shortening. This process stops the decomposition or rancidity of unsaturated fats. By controlling the degree of hydrogenation, it is possible to control the melting profile of the fats.
What is the product of bromination of cyclopentene?
For example, the bromination of cyclopentene would yield cis-1,2-cyclopentane and trans-1,2-cyclopentane.
What is the stereochemistry of the product formed when an alkene reacts with bromine solution?
The bromine atom in the bromonium ion acts as a shield in a way, forcing the bromonium anion to attack from the opposite side as it. … This is anti stereochemistry, which is defined as the two bromine atoms come from opposite faces of the double bond. The product is that the bromines add on trans to each other.
What is the product of the addition of cl2 to 2 butene?
Ernest Z. The product is meso because the intermediate involves anti addition to a cyclic chloronium ion.
What type of reagent is used for an epoxidation reaction?
Epoxidation is the chemical reaction which converts the carbon–carbon double bond into oxiranes (epoxides), using a variety of reagents including air oxidation, hypochlorous acid, hydrogen peroxide, and organic peracid (Fettes, 1964).
Can alkenes react with HCN?
Alkenes do add HCN across the double bond. The reaction is called hydrocyanation but the reaction mechanism is not an electrophilic addition.
What is hydrosilylation reaction?
From Wikipedia, the free encyclopedia. Hydrosilylation, also called catalytic hydrosilation, describes the addition of Si-H bonds across unsaturated bonds. Ordinarily the reaction is conducted catalytically and usually the substrates are unsaturated organic compounds.
What is the stereochemistry of hydrogenation?
The stereochemistry arises because both the hydrogen and the alkene must be adsorbed on the surface of a solid catalyst. The catalytic hydrogenation of an alkene results in a syn addition. … Then the alkene is adsorbed onto the metal surface. The H atoms add to the alkene while it is attached to the metal surface.
Why is hydrogenation called reduction?
Hydrogenation is a type of reduction reaction. It is used to convert unsaturated compounds into saturated compounds. Reduction refers to decreasing the oxidation number of a chemical species. It always occurs parallel to an oxidation reaction.
What is the structure of 3 3 Dimethylcyclopentene?
Chemical FormulaC7H12Molecular Weight96.17018 g/molIUPAC Name3,3-dimethylcyclopent-1-eneSMILES StringCC1(C)CCC=C1InChIInChI=1S/C7H12/c1-7(2)5-3-4-6-7/h3,5H,4,6H2,1-2H3
What are the product characteristics of hydrogenated fat?
Fully hydrogenated fats, processed in the same way, contain virtually no trans fats. The resulting product is firmer than its partially hydrogenated counterpart and has a hard, waxy consistency, even at room temperature.
What are the products of the acid catalyzed hydrolysis of a fat?
Fats and oils are esters of triglycerols and fatty acids. Hydrolysis can break down a fat or oil and release the triglycerol and fatty acids.