What is distillation organic chemistry
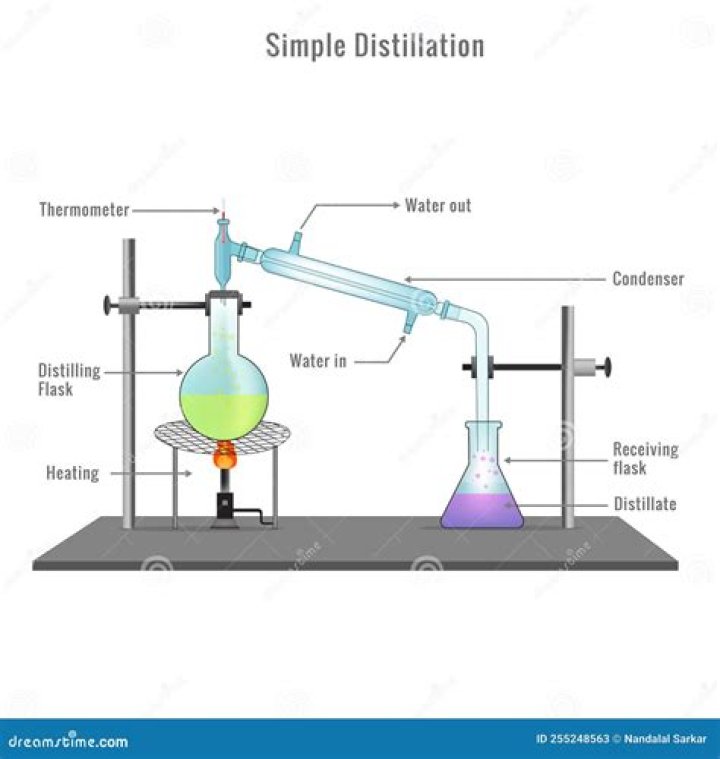
Distillation is a process of separating the component substances from a liquid mixture by selective evaporation and condensation. It is one of the most common laboratory techniques used by chemists for the purification and identification of organic liquids.
What is distillation in chemistry simple?
Simple distillation is a method for separating the solvent from a solution. For example, water can be separated from salt solution by simple distillation. … When the solution is heated, the water evaporates. It is then cooled and condensed into a separate container.
What is distillation explain?
Distillation, or classical distillation, is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation. Dry distillation is the heating of solid materials to produce gaseous products (which may condense into liquids or solids).
What is the purpose of simple distillation in organic chemistry?
Distillation is used to purify a compound by separating it from a non-volatile or less-volatile material. Because different compounds often have different boiling points, the components often separate from a mixture when the mixture is distilled.Where is distillation used?
Distillation is the process of separating components of a mixture based on different boiling points. Examples of uses of distillation include purification of alcohol, desalination, crude oil refining, and making liquefied gases from air.
What is distillation used for in chemistry?
Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or in the separation of two or more liquids having different boiling points, as in the separation of gasoline, kerosene, and lubricating oil from crude oil.
What processes are in distillation?
Distillation Process The process of distillation begins with heating a liquid to boiling point. The liquid evaporates, forming a vapor. The vapor is then cooled, usually by passing it through pipes or tubes at a lower temperature. The cooled vapor then condenses, forming a distillate.
What is the importance of simple distillation?
The most common purpose for simple distillation is to purify drinking water of unwanted chemicals and minerals such as salt. There are a variety of machines that distill liquids for the purpose of purification or alteration.What is the importance of distillation?
The distillation process is used to separate components based on their boiling points. Chemicals like common gas, diesel, and jet fuel achieve their boiling points at different temperatures. Distillation columns are used to separate mixed feed streams into their own distinct products.
What is distillation and their types?Distillation is the process in which water vapours converts into volatile liquids and separate through selective boiling. It is carried out between liquids having different boiling points. If we distillate again the pure liquid to increase the purity of liquid then the process is Double Distillation. …
Article first time published onWhat is distillation chemical engineering?
Distillation is simply defined as a process in which a liquid or vapor mixture of two or more substances is separated into its component fractions of desired purity, by the application and removal of heat. … Hence, when this vapor is cooled and condensed, the condensate will contain more volatile components.
What are distillation products?
Mid-range products include jet fuel, kerosene, and distillates (such as home heating oil and diesel fuel). … The heaviest products such as residual fuel oil are recovered at temperatures sometimes over 1,000 degrees Fahrenheit.
How is alcohol made by distillation?
Distillation Distillation is the process of separating alcohol from water via evaporation and condensation. The base alcohol is heated, and certain parts of it are captured. This process purifies and concentrates the remaining alcohol, which will ultimately be the final spirit produced. Distillation is done in stills.
How is alcohol made from distillation?
With pot distillation, you put a batch of fermented liquid (the beer or wine that you’re planning to distill) into a copper pot. You cap and seal the pot and heat it. As the liquid heats up, the alcohol in the liquid boils first (because alcohol boils at a lower temperature than water does) and turns to vapor.
What is evaporation chemistry?
evaporation, process by which an element or compound transitions from its liquid state to its gaseous state below the temperature at which it boils; in particular, the process by which liquid water enters the atmosphere as water vapour in the water cycle.
What is difference between fermentation and distillation?
Fermentation is where all alcohol is created, distillation is where the alcohol is separated and removed. In order for fermentation to occur, two things are needed: a raw material in liquid form that contains sugar, followed by the addition of yeast.
What is the basic principle of distillation?
Principle of Distillation It is heating the liquid to form a vapour and then cooling it to get back the liquid. It is used to separate components of a mixture containing two miscible liquids that boil without decomposition and have sufficient difference in their boiling points.
How is distillation used in the real world?
Distillation is used to separate mixtures of liquids by exploiting differences in the boiling points of the different components. The technique is widely used in industry, for example in the manufacture and purification of nitrogen, oxygen and the rare gases.
What is distillation Slideshare?
“Distillation is a process of separating the component substances from a liquid mixture by selective evaporation and condensation.” “Distillation is defined as the separation of the components of a liquid mixture by a process involving vaporization and subsequent condensation at another place.”
What is distillation column in chemistry?
In chemical analysis: Distillation. A distillation column is a tube that provides surfaces on which condensations and vaporizations can occur before the gas enters the condenser in order to concentrate the more volatile liquid in the first fractions and the less volatile components in the later fractions.
What is simple distillation example?
One example of a simple distillation mixture is separating saltwater to create pure water and salt. … Since sugar and water have different boiling points, the water will begin to evaporate first, and then cool to turn into pure water.
What is distillation in wine making?
Distillation is a process by which a low proof alcoholic beverage is heated in a controlled process in order to separate ethanol from water and other components, with the ultimate goal of concentrating the ethanol into a high proof spirit (Berglund, 2004).
Can you drink distilled water?
Distilled water is safe to drink. But you’ll probably find it flat or bland. That’s because it’s stripped of important minerals like calcium, sodium, and magnesium that give tap water its familiar flavor. What’s left is just hydrogen and oxygen and nothing else.
How do you make fermented alcohol?
It works like this: Pick a juice with at least 20g of sugar per serving, add a packet of specially designed yeast, plug the bottle with an airlock, and wait 48 hours. Just like the fermentation process used in winemaking, the juice’s natural sugar is converted into ethanol, with a byproduct of carbon dioxide.
How does distillation purify alcohol?
Separating alcohol from water can be done in several different ways, including distillation and freezing. The easiest way to separate alcohol and water is to distill it. This is done by heating the alcoholic mix in a round-bottom flask, then using a condensing column to trap the alcohol that evaporates from the water.
How do you distill water?
The process of distilling is simple. Heat tap water to the point that it turns to vapor. When the vapor condenses back to water, it leaves behind any mineral residue. The resulting condensed liquid is distilled water.
Why is distillation an effective way of separating alcohol and water?
Fractional distillation is a method for separating a liquid from a mixture of two or more liquids. For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points.