What is a concentration gradient Bozeman
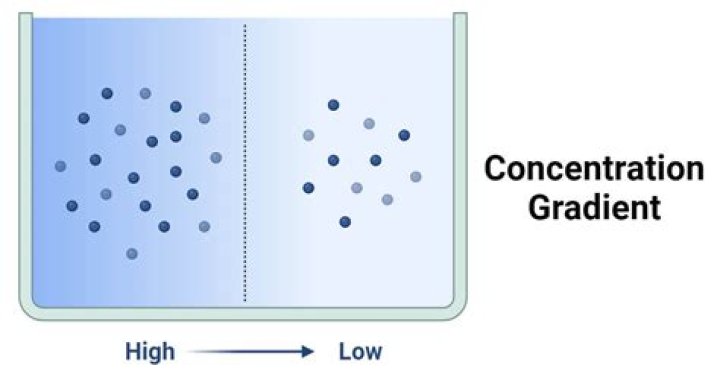
Concentration gradient refers to the gradual change in the concentration of solutes in a solution as a function of distance through a solution. … This imbalance of solutes between the two solutions drives solutes to move from a highly dense area to a lesser dense area.
What is a concentration gradient your answer?
Concentration gradient refers to the gradual change in the concentration of solutes in a solution as a function of distance through a solution. … This imbalance of solutes between the two solutions drives solutes to move from a highly dense area to a lesser dense area.
What is the concentration gradient of osmosis?
Osmosis occurs according to the concentration gradient of water across the membrane, which is inversely proportional to the concentration of solutes. Osmosis occurs until the concentration gradient of water goes to zero or until the hydrostatic pressure of the water balances the osmotic pressure.
What is a concentration gradient in cells?
The formal definition of a concentration gradient is the process of particles, which are sometimes called solutes, moving through a solution or gas from an area with a higher number of particles to an area with a lower number of particles. The areas are typically separated by a membrane.How does concentration gradient affect the rate of osmosis?
Concentration gradient – The movement of osmosis is affected by the concentration gradient; the lower the concentration of the solute within a solvent, the faster osmosis will occur in that solvent. Light and dark – They are also factors of osmosis; since the brighter the light, the faster osmosis takes place.
How does concentration gradient affect the rate of diffusion?
Extent of the concentration gradient: The greater the difference in concentration, the more rapid the diffusion. … Lower temperatures decrease the energy of the molecules, thus decreasing the rate of diffusion. Solvent density: As the density of a solvent increases, the rate of diffusion decreases.
What's an example of osmosis?
Examples of Osmosis: Examples of osmosis include red blood cells swelling up when exposed to fresh water and plant root hairs taking up water. To see an easy demonstration of osmosis, soak gummy candies in water. The gel of the candies acts as a semipermeable membrane.
What is the role of a concentration gradient in diffusion?
The concentration gradient therefore represents the concept that, just as a ball rolls down a slope, during diffusion molecules move down the concentration gradient. Higher concentration gradients will result in higher rates of diffusion. As the molecules move the gradient evens out until equilibrium is reached.Why do concentration gradients exist?
A concentration gradient exists when a higher concentration of a solute is separated from a lower concentration, by a semipermeable membrane.
What does it mean to move down the concentration gradient?Down a Concentration Gradient refers to going from regions of high concentration of some entity to regions of low concentration, and such movement generally occurs spontaneously, that is, if allowed to happen it happens.
Article first time published onWhat is dC DX in calculus?
Calculus allows you to calculate infinitely many gradients that are each infinitesimally short. … So the gradient is dC/dx (which, you may recall, is a “derivative”) . dC/dx tells you how much the concentration changes as you move. The gradient is measured in units of M m-1 or M cm-1.
What is the difference between concentration gradient and electrical gradient?
Simple concentration gradients are differential concentrations of a substance across a space or a membrane, but in living systems, gradients are more complex. … The electrical gradient of K+, a positive ion, also tends to drive it into the cell, but the concentration gradient of K+ tends to drive K+ out of the cell.
What is a concentration gradient quizlet?
The concentration gradient is the gradual difference in the concentration of solutes in a solution between two regions.
How does tonicity affect the function of the cell?
The tonicity of a solution is related to its effect on the volume of a cell. A hypotonic solution causes a cell to swell, whereas a hypertonic solution causes a cell to shrink. … Although it is related to osmolality, tonicity also takes into consideration the ability of the solute to cross the cell membrane.
What must happen when a concentration gradient is eliminated?
Diffusion will continue until the concentration gradient has been eliminated. … These molecules diffuse freely in and out of the cell, along their concentration gradient. Though water is a polar molecule, it can also diffuse through the plasma membrane.
What does Crenation mean and why does it occur?
In biology, crenation describes the formation of abnormal notched surfaces on cells as a result of water loss through osmosis. … The cells start to shrivel and form abnormal spikes and notches on the cell membrane. This process is called crenation.
How does solute concentration affect the movement of water?
The volume on the side with the solute increases until the number of water molecules on both sides is equal. Increasing the concentration of solute reduces the space available for water molecules, which reduces their numbers. This in turn increases the tendency of the water to flow into that side from the other side.
How do you explain osmosis to a child?
Osmosis allows water to pass through cell membranes. The transfer of water from a dilute solution to a concentrated solution over a partly permeable membrane is defined as osmosis.
Is Sweating an example of osmosis?
Potato Osmosis Lab Report Your sweat glands use osmosis. Instead it deposits a little bit of salt inside one of you sweat glands.
What is osmosis diagram?
Osmosis is the diffusion of water across a partially permeable membrane from a dilute solution (high concentration of water) to a concentrated solution (low concentration of water). In the diagram, the concentration of sugar is initially higher on the right side of the membrane.
How does concentration gradient affect the rate of diffusion GCSE?
The rate of diffusion is affected by the: Concentration gradient – a larger difference in the high and low concentrations increases the diffusion rate. Temperature – a higher temperature provides the particles with more kinetic energy increasing the diffusion rate.
Why do particles move down a concentration gradient?
A concentration gradient occurs when the concentration of particles is higher in one area than another. In passive transport, particles will diffuse down a concentration gradient, from areas of higher concentration to areas of lower concentration, until they are evenly spaced.
What is the meaning of higher concentration and lower concentration?
High concentration means the amount of solute is high in the solution and low concentration means it is low. Amount of solvent is high in low concentration and low in high concentration of solute.
Why are concentration gradients important for organisms?
Concentration gradients are used by many cells to complete a wide variety of tasks. In fact, there is energy stored in a concentration gradient because the molecules want to reach equilibrium. So, this energy can be utilized to accomplish tasks.
How are concentration gradients created within a cell?
Terms in this set (30) How are concentration gradients created within a cell? The plasma membrane is semi-permeable. The arrangement of amphipathic phospholipids into a bilayer prevents most substances from being permeable without the help of channels and transporters.
Why are gradients important in diffusion and osmosis?
How are gradients important in diffusion and osmosis? Gradients allow materials to move across membranes, from an area of high concentration to an area of low concentration. … Water will move out of a plant cell if it has a higher water potential (concentration) than the surrounding environment.
How does concentration gradient represent potential energy?
The concentration gradient of a substance across a membrane represents potential energy because it drives diffusion.
How does the concentration gradient affect the process of ATP synthesis?
The proton gradient produced by proton pumping during the electron transport chain is used to synthesize ATP. Protons flow down their concentration gradient into the matrix through the membrane protein ATP synthase, causing it to spin (like a water wheel) and catalyze conversion of ADP to ATP.
How is concentration gradient related to the rate at which simple diffusion occurs?
Simple passive diffusion occurs when small molecules pass through the lipid bilayer of a cell membrane. … Since the net movement of diffusing molecules depends on the concentration gradient, the rate of diffusion is directly proportional to the concentration gradient (dC/dx) across the membrane.
Why can't Nemo live in a tank with goldfish?
why can’t nemo live in a fish tank filled with goldfish? Memo’s cells are hypertonic to the freshwater, so water would move into his cells and cause them to burst. what would happen to your cells if you tried to drink ocean water?
What is the difference between down a concentration gradient and against a concentration gradient?
Since diffusion moves materials from an area of higher concentration to an area of lower concentration, it is described as moving solutes “down the concentration gradient” (compared with active transport, which often moves material from area of low concentration to area of higher concentration, and therefore referred …