What can Decolourise aqueous bromine
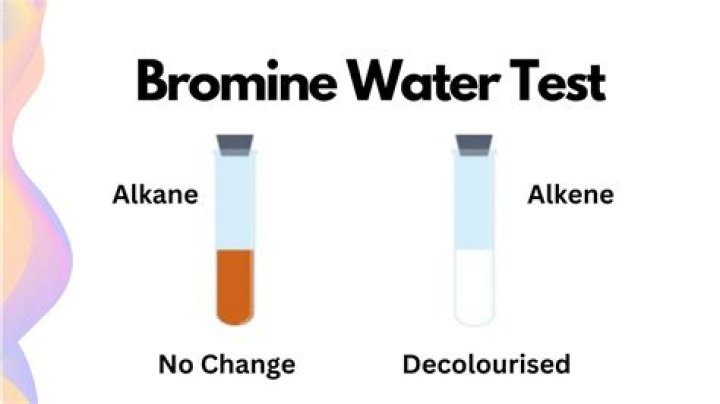
Alkenes can decolourise bromine water
What causes bromine to Decolourise?
An alkene decolourise bromine water because the bromine reacts with the carbon-carbon double bonds. The carbon-carbon bond is broken and bromine gets attached to the alkene thus forming alkane.
Which compound will Decolourise bromine water the fastest?
Answer: Alkene decolorizes bromine water so it is often used as a test for unsaturated hydrocarbon.
What reacts with aqueous bromine?
The most common compounds that react well with bromine water are phenols, alkenes, enols, the acetyl group, aniline, and glucose. … In this reaction as well the color of bromine water is changed to colorless from yellow (oxidation process).Which hydrocarbons can Decolourise bromine water?
C6H12 (Hexene) and C6H10 (Hexyne or Cyclohexene ) are the hydrocarbons which can decolourize bromine water, as they are unsaturated. Bromine undergo addition reactions with unsaturated compounds like alkenes and alkynes containing a double or a triple bond.
Does benzene Decolourise bromine water?
Benzene does not decolourise bromine water.
Is aqueous bromine polar?
Br- , bromide ion is as polar as can be , as it is a mononegative anion. As as an unstable intermediate, Br the atom would be nonpolar free radical . According to the electronegativity values, bromine is more electronegative (2.9) than iodine (2.7).
Is bromine aqueous or liquid?
Bromine is a dark reddish-brown fuming liquid with a pungent odor. Denser than water and soluble in water.What is a molecule of bromine?
Elemental bromine is a diatomic molecule (Br 2 ). Bromine will combine with most other elements. Reaction with metallic elements leads to salts such as silver bromide (AgBr), in which the bromine atom has a −1 charge and oxidation number.
Does phenol Decolourise bromine water?Hint: When bromine water is added to a solution of phenol in water, the bromine water is decolourised and a white precipitate is formed which smells of antiseptic. … We know that the reaction of phenol and water with bromine is known as bromination of phenol.
Article first time published onCan you Decolourise bromine water?
Bromine water can not decolourize them. Alkanes are hydrocarbons that are saturated and therefore do not decolorize bromine water.
Does ketones Decolourise bromine water?
Explanation: yes aldehydes and ketone decolourises bromine water because they are unsaturated.
Which of the following will not Decolourise bromine water?
C3H8 (propane) is an alkane (saturated hydrocarbon) and hence it will not decolorize bromine water.
What compounds Decolourise bromine?
Ethene, butyne, and hexyne can decolourize bromine water. The bromine loses its unique red-earthy colored tone to give a dry fluid. The decoloration of bromine is frequently utilized as a test for $C – C$ twofold and triple bond.
Does aldehyde Decolourise bromine water?
The answer is yes. Aldehydes will decolourise bromine water.
Which of the following will Decolourise bromine water 1 point c4h10 C6H14 C6H12 c8h18?
C6H12 and C6H10 an decolourise bromine water since these are unsaturated hydrocabons. C6H14 cannot decolourise bromine water since it is a saturated hydrocarbon.
Is liquid bromine polar or nonpolar?
Elemental bromine has two bromine atoms covalently bonded to each other. Because the atoms on either side of the covalent bond are the same, the electrons in the covalent bond are shared equally, and the bond is a nonpolar covalent bond.
Is bromine insoluble in non polar solvents?
Explanation: Bromine is a nonpolar solvent. Only nonpolar molecules will be soluble in or miscible with bromine. Thus, only the following nonpolar molecules will be soluble in or miscible with bromine: C5H12 (l), vegetable oil, and I2 (s).
Is H2O polar or nonpolar?
Water (H2O), like hydrogen fluoride (HF), is a polar covalent molecule. When you look at a diagram of water (see Fig. 3-2), you can see that the two hydrogen atoms are not evenly distributed around the oxygen atom.
Does alkane Decolourise bromine water?
Bromine water is an orange solution of bromine. It becomes colourless when it is shaken with an alkene. Alkenes can decolourise bromine water, but alkanes cannot.
Does toluene Decolourise bromine water?
Why doesn’t toluene decolourise the reddish brown solution of Br2/CCl4? – Quora. A molecule of toluene (methylbenzene) contains two parts: a benzene ring (-C₆H₅) and a methyl group (-CH₃). the methyl group does not undergo radical substitution with bromine as there is no ultraviolet light to initiate the reaction.
Does benzene Decolourise kmno4?
Benzene does not decolorize the alkaline permanganate solution due to the delocalization of pi electrons. Also, it is resonance stabilized and doesn’t react with an alkaline permanganate solution.
What is liquid bromine?
What bromine is. Bromine is a naturally occurring element that is a liquid at room temperature. It has a brownish-red color with a bleach-like odor, and it dissolves in water.
Is bromine a molecule or atom?
Diatomic bromine is an element with a red-brown color and offensive odor discovered by Antoine-Jerome Balard in 1826. It is one of two elements that is a liquid at room temperature. In its pure form, it exists as a diatomic molecule and can be toxic to human health.
What are chemical properties of bromine?
atomic number35melting point−7.2 °C (19 °F)boiling point59 °C (138 °F)specific gravity3.12 at 20 °C (68 °F)oxidation states−1, +1, +3, +5, +7
Can bromine be aqueous?
Bromine: It is soluble in water. Its aqueous solution is called bromine water.
Is aqueous bromine soluble in water?
Bromine is a dense, mobile, fuming, reddish-brown liquid at room temperature. … Bromine is slightly soluble in water and highly soluble in many organic solvents, including carbon disulfide, carbon tetrachloride, acetic acid, and aliphatic alcohols.
Is bromine a volatile liquid?
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest halogen, and is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour.
How does phenol react with aqueous bromine?
If bromine water is added to a solution of phenol in water, the bromine water is decolorized and a white precipitate is formed which smells of antiseptic. The precipitate is 2,4,6-tribromophenol.
Does aniline Decolourise bromine water?
When aniline treated with bromine water, the bromine water gets decolourized and white precipitate is formed. This reaction results in the formation of 2,4,6-tribromo phenylamine. This reaction is an example of bromination.
Why does phenol Decolourise bromine water but not benzene?
Benzene does not undergo “adding across the multiple bond” reactions which would destroy the delocalization. Benzene does undergo substitution reactions. If the reaction of benzene with bromine could produce phenyl bromide, the bromine water would be decolorized.