How many protons does gallium have
The nucleus consists of 31 protons (red) and 38 neutrons (blue). 31 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The stability of an element’s outer electrons determines its chemical and physical properties.
How many protons and electrons are in gallium?
The nucleus consists of 31 protons (red) and 38 neutrons (blue). 31 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The stability of an element’s outer electrons determines its chemical and physical properties.
How many protons would gallium 72 have?
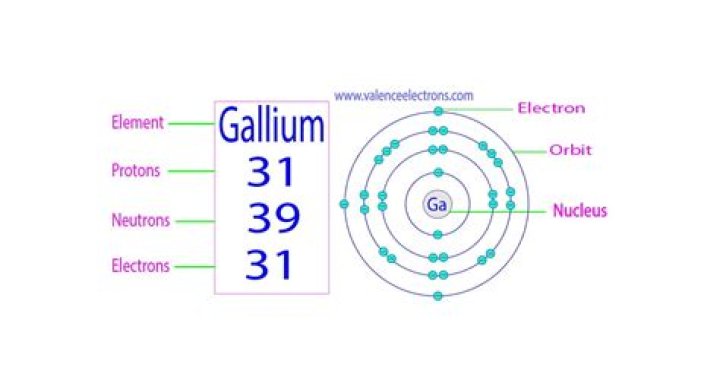
Atomic Number of Gallium Gallium is a chemical element with atomic number 31 which means there are 31 protons and 31 electrons in the atomic structure.
How many protons are there in gallium 71?
Properties of Gallium-71 Isotope:GALLIUM-71Nucleon Number (A)71Proton Number (Z)31Half-lifeStableSpin1.5How many protons does Gallium 69 have?
Properties of Gallium-69 Isotope:GALLIUM-69Nucleon Number (A)69Proton Number (Z)31Half-lifeStableSpin1.5
How many protons does Gallium 68?
Element NameGalliumElement SymbolGaAtomic Number31
How many protons does Gallium 70?
Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of gallium-70 (atomic number: 31), an isotope of this element. The nucleus consists of 31 protons (red) and 39 neutrons (orange).
How many electrons does Gallium 71 have?
Explanation: Gallium, Ga has 31 protons and 31 electrons. The superscripts represent the electrons present in each region of the periodic table. The sum of these superscripts should equal the atomic number for a neutral atom.How many protons does Titanium 48 have?
Properties of Titanium-48 Isotope:TITANIUM-48Atomic Number (Z)22Mass Number (A)48Nucleon Number (A)48Proton Number (Z)22
What element has 78 protons and 92 neutrons?Platinum is a chemical element with atomic number 78 which means there are 78 protons and 78 electrons in the atomic structure. The chemical symbol for Platinum is Pt.
Article first time published onHow many protons does arsenic 75?
To get its mass number, you need to round its atomic mass to the nearest whole number. Therefore, the stable arsenic isotope is arsenic-75, 75As , which has 33 protons and 42 neutrons in its nucleus.
How many electrons does Gallium 67 have?
Gallium atoms have 31 electrons and the shell structure is 2.8. 18.3.
How many neutrons are in CA?
Johnson Z. In calcium-40, there are 20 neutrons.
How many nucleons does gallium have?
Now for gallium, Z=31 , i.e. in each and every gallium nucleus there are 31 massive, positively charged particles; this is what defines the element as gallium. And so in 69Ga , there must be 69−31=38⋅neutrons ….and in 71Ga , there must be 71−31=40⋅neutrons ….
How many neutrons are in gallium 68?
NuclideZNExcitation energy68Ga313769Ga313870Ga3139
What are some examples of protons?
Examples of Protons The nucleus of a hydrogen atom or the H+ ion is an example of a proton. Regardless of the isotope, each atom of hydrogen has 1 proton; each helium atom contains 2 protons; each lithium atom contains 3 protons and so on.
What are protons equal to?
The number protons equals the number of electrons in a NEUTRAL atom.
What is proton and neutron?
Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).
What is Gallium 70 mass number?
PubChem CID6337546StructureFind Similar StructuresMolecular FormulaGaSynonymsGallium-70 Gallium, isotope of mass 70 70Ga 14391-74-3Molecular Weight69.92602
How many neutrons are in Argon?
There are 22 neutrons in one atom of argon. On the periodic table, argon’s atomic number is 18. This tells you that it has 18 protons.
What is the mass of gallium 70?
The atomic mass of gallium is 69.72 amu.
Is gallium 62 stable?
No, it would not be a stable isotope. According to the graph, Gallium (which has 31 protons) does not have a known isotope with 31 neutrons.
Who discovered GA 68?
Gallium, discovered in 1875 by Paul-Émile Lecoq de Boisbaudran, was first considered for diagnostic and therapeutic medical use in the 1940s by the research of H. C. Dudley and his co-workers.
What is Gallium 67 used for in medicine?
Gallium citrate Ga 67 injection is used to help your doctor diagnose if you have certain types of cancer, such as Hodgkin’s disease, lymphoma, or lung cancer. It is also used to help your doctor see if you have acute swollen lesions.
How many protons are in calcium 41?
half-life (λ)100,000yProtons20Neutrons21
How many protons are in Li?
This means that lithium has 3 protons, 3 electrons and 4 neutrons (6.941 – 3 = ~4). Being an alkali metal, lithium is a soft, flammable, and highly reactive metal that tends to form hydroxides.
How many protons neutrons and electrons does TI 47 have?
(physics) A minor stable isotope of titanium, 47 22Ti, having twenty-two protons and twenty-five neutrons; it amounts to about 7% of the element in nature.
How do u find neutrons?
To find the number of neutrons, subtract the number of protons from the mass number. number of neutrons=40−19=21.
How many protons neutrons and electrons does germanium have?
Diagram of the nuclear composition and electron configuration of an atom of germanium-74 (atomic number: 32), the most common isotope of this element. The nucleus consists of 32 protons (red) and 42 neutrons (blue). 32 electrons (green) bind to the nucleus, successively occupying available electron shells (rings).
How many neutrons are in the radioactive isotope 40 19k?
They all have 19 protons, but they have different numbers of neutrons. K has 20 neutrons, 4019K has 21 neutrons, and 4119K has 22 neutrons. 4 Naturally occurring iodine is iodine-127. The radioactive isotopes of iodine-125 and idine-130 are used in nuclear medicine.
What is the element that has 20 protons?
Ca has 20 protons, so neutral it would have 20 electrons, but according to the charge, 2 electrons have been lost.