How many electrons will go in the first shell of sodium
So, sodium’s 11 electrons are arranged this way: 2 electrons in the first “shell”, 8 electrons in the second “shell”; and 1 electron (the valence electron) in the third “shell”. We write this as 2.8. 1.
How many electrons will go in the first shell of a sodium atom?
So… for the element of SODIUM, you already know that the atomic number tells you the number of electrons. That means there are 11 electrons in a sodium atom. Looking at the picture, you can see there are two electrons in shell one, eight in shell two, and only one in shell three.
How many electrons will go in the third shell of sodium?
There is only one electron in the third shell of a neutral sodium atom. We must make the distinction that it is a neutral sodium atom because sodium…
How many electrons are in each shell for sodium?
ZElementNo. of electrons/shell11Sodium2, 8, 112Magnesium2, 8, 213Aluminium2, 8, 314Silicon2, 8, 4How many electrons does sodium have?
Each element has its own atomic number. The atomic number of an element tells you how many protons that the element has. This is written at the bottom left hand side of the symbol. Since atoms are neutral, we know then that sodium atoms must also have 11 negative electrons to cancel the charge from 11 positive protons.
How many valence electrons are in Na+?
The positive charge dictates all the properties of Na+ , since otherwise it is stable and inert with its eight electrons in the valence shell. A sodium atom, would strive to lose its lone valence shell electron.
How many shells are in a sodium ion?
Forming positive ions A sodium atom has one electron in its outer shell. The atom is more stable if it has a full outer shell. A sodium atom can lose its outer electron. It will still have 11 positive protons but only 10 negative electrons.
How do you find electrons in shells?
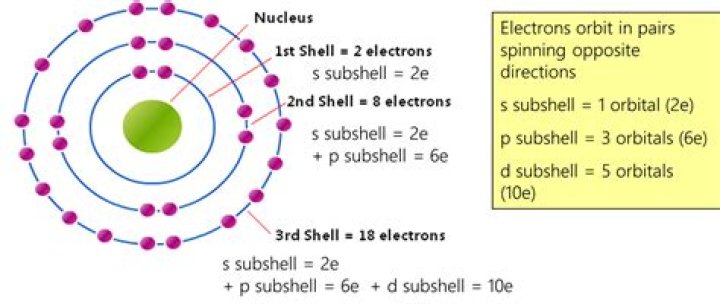
Rule 1: The maximum number of electrons present in a particular shell is calculated by the formula 2n2, where “n” represents the shell number. For instance, K shell is the first shell and it can hold up to 2(1)2 = 2 electrons. Similarly, L shell is the second shell and it can hold up to 2(2)2 = 8 electrons.Does the third shell have 8 or 18 electrons?
In this sense the third shell can hold 8 electrons. 4s2 not the third shell, but the next 10 electrons go into the 3d orbitals that are part of the third shell but shown on the fourth shell level. … So the third shell can be considered to hold 8 or 18 electrons but in total the third shell can hold 18 electrons.
Is sodium outer shell complete?Sodium, with its single outermost electron, reacts strongly and forms highly stable compounds with elements that need a single electron to complete their outermost shell. … The sodium atom is left with a full outermost electron shell with eight electrons, and the outermost shell of the other atom is full as well.
Article first time published onHow do you find the electrons?
- The number of protons in the nucleus of the atom is equal to the atomic number (Z).
- The number of electrons in a neutral atom is equal to the number of protons.
Does sodium have 1 valence electrons?
A: An atom of a group 1 element such as sodium has just one valence electron. It is “eager” to give up this electron in order to have a full outer energy level, because this will give it the most stable arrangement of electrons.
How many electrons go in the 4th shell?
n=ShellMaximum Number of Electrons22nd Shell833rd Shell1844th Shell32
What is the 2-8-8 rule in chemistry?
There is a 2-8-8 rule for these elements. The first shell is filled with 2 electrons, the second is filled with 8 electrons, and the third is filled with 8. You can see that sodium (Na) and magnesium (Mg) have a couple of extra electrons. They, like all atoms, want to be happy.
How many electrons are in the fourth shell?
The fourth shell holds 32 electrons; 2 in a 4s orbital; 6 in three 4p orbitals; 10 in five 4d orbitals; and 14 in seven 4f orbitals. The exact arrangement of electrons in an atom’s shells and subshells is the atom’s electron configuration.
How many outer shell electrons does group 1 have?
The atoms of all group 1 elements have similar chemical properties and reactions because they all have one electron in their outer shell. Similarly, the atoms of all group 7 elements have similar chemical properties and reactions to each other, because they all have seven electrons in their outer shell.
How many electrons are in the outer shell of Group 7?
The Group 7 elements – fluorine (F), chlorine (Cl), bromine (Br), iodine (I) and astatine (At) – have seven electrons in the outer shell.
What is an outer shell electron?
The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons.
How many electrons will go in the first shell of chlorine?
So… for the element of CHLORINE, you already know that the atomic number tells you the number of electrons. That means there are 17 electrons in a chlorine atom. Looking at the picture, you can see there are two electrons in shell one, eight in shell two, and seven in shell three.
How many electron shells does boron have?
Atomic Number5Mass Number11Number of Neutrons6Shell structure (Electrons per energy level)[2, 3]
How many electrons does sodium need to fill its outer shell?
As Figure 1 illustrates, sodium (Na) only has one electron in its outer electron shell. It takes less energy for sodium to donate that one electron than it does to accept seven more electrons to fill the outer shell.
How many electrons are there in Na+ ions if the atomic number of Na atom is 11?
There are 10 electrons present in Na+. The atom of sodium has 11 electrons, 11 protons along with 12 neutrons, but Na+ contains one less electron, 11 protons along with 12 neutrons, as the ion has lost 1 electron.
How many electrons are in the outer shell of the Na ion?
The outermost shell of the sodium ion is the second electron shell, which has eight electrons in it.
How many electrons are in each shell?
Energy shellMaximum number of electronsFirst2Second8Third8
How many atoms does sodium have?
This is because there is 1 atom of Na (sodium) and 1 atom of Cl (chlorine) in each NaCl molecule. Elements by themselves do not have a “number of atoms”- if you’re talking about the atomic number, it’s the number of protons (or electrons in a neutral atom) of an element.
What is the electron number?
Finding the Number of Electrons The number of electrons in an atom is equal to the atomic number of an element, for neutrally charged species. This means the number of electrons and the number of protons in an element are equal. Therefore, the number of electrons in oxygen is 8.
What is sodium valence shell?
Sodium has one valence electron. Valence electrons are electrons found in the outermost shell of an atom. The shell number representing the valence shell will differ depending on the atom in question. For sodium, which is in the 3rd row of the periodic table, the valence electrons will be found in the 3rd shell.
What number shell is the valence electron shell of a sodium atom what number shell is the valence shell of a sodium ion explain the difference?
For sodium, the valence shell is the third shell; for the sodium ion, the valence shell is the second shell because it has lost all its third shell electrons. The valence shell for both bromine atom and bromide ion is n=4.