How is the concentration of the calcium determined
Calcium and magnesium ions dissolved in water cause water hardness. … In order to determine the concentration of Ca2+ in water, the Mg2+ must be removed first, usually by precipitation. The titration with EDTA then determines the concentration of Ca2+ remaining in the water.
How do you find the concentration of calcium ions in a solution?
You can try calmagite colorimetric method suggested by Hash.com (Method 8030) to measure calcium ions in solution. However, this method is for low concentrations of Ca2+ (0.05 to 4 mg/l). It means that you need to dilute your solution at the time of measurement!
How is calcium concentration measured in water?
This can be determined using atomic absorption spectroscopy (AA) or inductively coupled plasma atomic emission spectroscopy (ICP). However, a much simpler way is by ionizing acid-bound calcium using acidizing pretreatment. The LAQUAtwin Ca2+ can be used to measure the total amount of calcium.
What is the concentration of calcium?
The total calcium concentration in the plasma is 4.5-5.1 mEq/L (9-10.2 mg/dL). Fifty percent of plasma calcium is ionized, 40% is bound to proteins (90% of which binds to albumin), and 10% circulates bound to anions (eg, phosphate, carbonate, citrate, lactate, sulfate).How do you measure calcium in water sample?
Calcium in water will be analyzed this week by EDTA titration and next week by atomic absorption analysis and the results compared. Water hardness can be readily determined by titration with the chelating agent EDTA (ethylenediaminetetraacetic acid).
How do you calculate calcium from calcium hardness?
To convert calcium as CaCO3 to the elemental form of just calcium, multiply the Ca hardness value by 0.4.
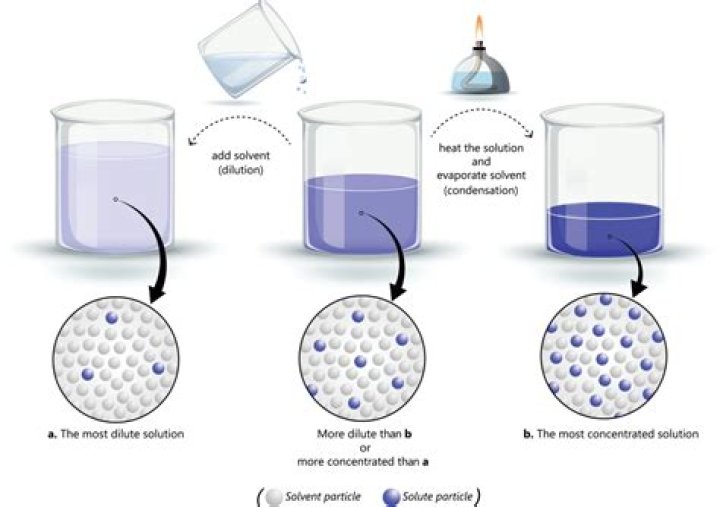
How do you determine concentration?
Divide the mass of the solute by the total volume of the solution. Write out the equation C = m/V, where m is the mass of the solute and V is the total volume of the solution. Plug in the values you found for the mass and volume, and divide them to find the concentration of your solution.
What regulates intracellular calcium concentration?
These data show that intracellular calcium in horizontal cells is regulated by voltage-dependent L- and N-type calcium channels, ionotropic AMPA and kainate receptors, and release of calcium from internal stores after activation of ryanodine receptors.How the concentration of calcium within the cell is increased?
In response to a chemical, electrical, or physical stimulus interaction with a cell surface receptor, intracellular calcium concentrations rise from an influx of extracellular calcium or from intracellular calcium stores such as the endoplasmic or sarcoplasmic reticulum.
What is the concentration of calcium ions in the endoplasmic reticulum?Whereas the ER lumen contains calcium ions in the high micromolar range (~100 µM), in a resting cell the cytosolic calcium concentration is low nanomolar (50–100 nM).
Article first time published onHow can you distinguish between concentration of Ca 2 and Mg 2 ions using titration procedure?
For the titration, the sample solution containing the calcium and magnesium ions is reacted with an excess of EDTA. The indicator is added and remains blue as all the Ca2+ and Mg2+ ions present are complexed with the EDTA. A back titration is carried out using a solution of magnesium chloride.
How do you calculate molar concentration of EDTA?
- Molarity.
- EDTA (mol / L)
- = 0.01000 L × 0.01603 mol / L ×
- 1 mol EDTA.
- 1 mol Zinc.
- ×
- 0.01625 L.
What is the concentration of calcium in milk?
Dairy and SoyAmountCalcium (mg)Milk (skim, low fat, whole)1 cup300Buttermilk1 cup300Cottage Cheese0.5 cup65Ice Cream or Ice Milk0.5 cup100
Which standard solution is used in standardization in Ca determination?
The classic method of determining calcium and other suitable cations is titration with a standardized solution of ethylenediaminetetraacetic acid (EDTA).
What are 3 ways to measure the concentration of a solution?
What are three ways to measure the concentration of a solution? Concentration can be expressed as percent by volume, percent by mass, and molarity.
How do you calculate the concentration of vitamin C?
Calculation. For standard solution Mass Ascorbic acid = Mole iodine × Volume of iodine × 176.12 = 0.00231 × 45 × 176.12 = 91.54 mg Initially, the amount of Ascorbic acid was taken for 100mg and therefore for total amount of ascorbic acid i.e. 250 mg the ratio stands out to be 2.5 (250/100).
How do you determine the degree of hardness?
The hardness of water is calculated in terms of the weight of $CaC{O_3}$. 1ppm = 1mg/L. The molecular weight of $CaC{O_3}$ = 100. The degree of hardness is expressed as parts per million (ppm).
How do I calculate ppm?
How do you calculate ppm? PPM is calculated by dividing the mass of the solute by the mass of the solution, then multiplying by 1,000,000. Both parts of the equation must be in the same format, weight or volume.
How do you calculate total hardness?
The total hardness is determined by the sum of Ca and Mg: TH = (2×1.40 + 2×0.23) meq/L = 3.26 meq/L – as presented in the diagram above.
What is homeostatic mechanism of calcium?
Calcium homeostasis is under the direct control of the parathyroid gland, PTH, calcitonin, and calcitriol, which is a vitamin D metabolite. The ionized form is the physiologically active form.
Is calcium concentration higher inside or outside the cell?
Calcium Pumps Calcium exists as a gradient across the plasma membrane, with extracellular concentrations being about 10,000 times higher than intracellular ones.
What are the mechanisms in place that keep the cytosolic Ca2+ concentration tightly regulated?
Normal extracellular calcium concentration is regulated by three calciotropic hormones: parathyroid hormone (PTH), calcitonin, and vitamin D. These hormones act on three target organs—kidney, bone, and intestine—to maintain serum calcium within a very narrow range.
Why is the regulation of Ca2+ so critical to homeostasis?
Why is the regulation of Ca2+ so critical to homeostasis? … Ca2+ is important in cardiac and skeletal muscle contraction.
How is calcium Signalling regulated and maintained?
SOCE is regulated by STIM1 which senses the ER lumen Ca2+ content via its EF-hands. Upon ER Ca2+ depletion, STIM1 undergo a Ca2+-regulated conformational change that promotes its oligomerization and activation of Orai Ca2+ channels.
What is the role of Ca2+ or Mg2+ in the physiological process?
Ca2+ and Mg2+ are essential ions in a wide variety of cellular processes and form a major constituent of bone. It is, therefore, essential that the balance of these ions is strictly maintained.
How intracellular Ca2+ concentration affect the cell Signalling?
Calcium signaling is the use of calcium ions (Ca2+) to communicate and drive intracellular processes often as a step in signal transduction. Ca2+ is important for cellular signalling, for once it enters the cytosol of the cytoplasm it exerts allosteric regulatory effects on many enzymes and proteins.
Which function is wrong WL Golgi?
– Aid in the formation of plant cell walls: Golgi bodies of plant cells synthesize all types of polysaccharides such as pectin, hemicellulose, and microfibrils of a-cellulose. Therefore the first statement (option a) regarding the function of the Golgi apparatus is incorrect.
What releases calcium ions from the endoplasmic reticulum?
When the muscle is stimulated, calcium ions are released from its store inside the sarcoplasmic reticulum, into the sarcoplasm (muscle ).
How do you identify calcium?
A calcium blood test does not tell you how much calcium is in your bones. Bone health can be measured with a type of x-ray called a bone density scan, or dexa scan. A dexa scan measures the mineral content, including calcium, and other aspects of your bones.
What is the confirmatory test for calcium?
Dissolved precipitate in acetic acid and boiled it. Added few drops of ammonium oxalate to above solution and then added ammonium hydroxide. White precipitate is formed. Ca(II) Confirmed.
How do you determine the purity of calcium hydroxide?
Dip the strip of the pH paper strip into the solution and then take it out. The pH paper will change its color. Compare the color of the paper with the scale enclosed with the pH paper to assign the pH of the solution. A basic pH value such as 10 to 11 is indicative for calcium hydroxide.