How does the iodine test for starch work
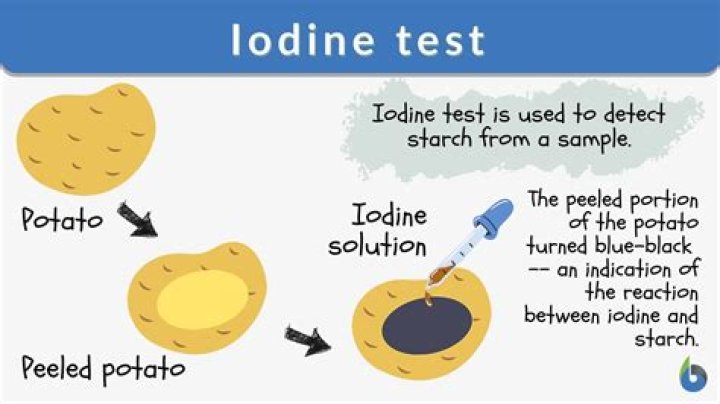
Using iodine to test for the presence of starch is a common experiment. A solution of iodine
How does the test for starch work?
A chemical test for starch is to add iodine solution (yellow/brown) and look for a colour change. In the presence of starch, iodine turns a blue/black colour. … The test involves heating a solution of the sugar to be tested with Benedict’s reagent and observing the colour change of blue to orange.
What happens when you add a drop of iodine to starch?
When two or more drops of iodine solution fall on starch substance, we observe a blue-black colouration. … In the presence of starch, this yellow colour changes to blue-black colour due to the formation of starch-iodine complex that imparts the blue colour.
How do you test for starch solution iodine?
To prepare starch indicator solution, add 1 gram of starch (either corn or potato) into 10 mL of distilled water, shake well, and pour into 100 mL of boiling, distilled water. Stir thoroughly and boil for a 1 minute. Leave to cool down. If the precipitate forms, decant the supernatant and use as the indicator solution.What is iodine method?
Iodine measurement in biological samples is carried out almost exclusively by one of two methods: One is a kinetic spectrophotometric method called the Sandell–Kolthoff reaction based on the reduction of yellow Ce(IV) by As(III) to colorless Ce(III), which is normally very slow.
What is the composition of iodine test?
The reagent used in the iodine test is Lugol’s iodine, which is an aqueous solution of elemental iodine and potassium iodide. Iodine on its own is insoluble in water.
How do the results of the iodine test indicate that hydrolysis of starch occurred?
The amylose in starch forms helices where iodine molecules assemble, forming a dark blue or black color. When starch is broken down or hydrolyzed into smaller carbohydrate units, the blue-black color is not produced. Therefore, this test can also indicate completion of hydrolysis when a color change does not occur.
Which of the following would show positive test for the presence of starch?
Bread will show positive test for the presence of starch. It will turn the iodine solution to blueish black.Why is iodine solution used to test the presence of starch in a leaf?
When you put iodine on the leaves, one of them will turn blue-black and the other will be a reddish-brown. Iodine is an indicator that turns blue-black in the presence of starch. The leaf that was in the light turns blue-black, which demonstrates that the leaf has been performing photosynthesis and producing starch.
What happened after adding iodine to both test tubes A and B?Dilute Iodine solution was added to the test tube. 5. In test tube A, the Iodine solution color remained the same which indicates the absence of starch. … In test tube B, the Solution turns into a black due to the presence of Starch.
Article first time published onHow do you analyze iodine?
The quantities of iodine within the body can be assessed through the use of bioassays that are comprised of in vivo measurements and/or in vitro measurements. In vivo measurements can be obtained through techniques that directly quantify internally-deposited iodine using, for example, thyroid or whole body counters.
How do you measure iodine?
Iodine value (IV) is a measure of the unsaturation of oil and fat. It is expressed as the mass of iodine consumed by 100 g of oil or fat, and it is usually determined by chemical methods such as Wijs method, Hanus and Hubel, Hofmann and Green, Rosenmund-Kuhnhenn, or a modification of the Wijs method.
How is iodine number determined?
It is defined as the amount of iodine (in g) added to 100 g of oil. Obviously, the iodine content can be easily calculated by analyzing the composition in fatty acids. In this case, the percentage of each unsaturated fatty acid is multiplied by a preestablished constant and all results are summed.
How can you tell when the hydrolysis of starch is complete Why does the test work this way?
When starch is hydrolyzed and broken down to small carbohydrate units, the iodine will not give a dark blue (or purple) color. The iodine test is used in this experiment to indicate the completion of the hydrolysis.
What is indicated by the colored result in iodine test for starch?
Iodine Test A solution of iodine (I2) and potassium iodide (KI) in water has a light orange-brown color. If it is added to a sample that contains starch, such as the bread pictured above, the color changes to a deep blue. But how does this color change work? Starch is a carbohydrate found in plants.
Why is starch test done?
Hint: Starch test is done to find out the presence of carbohydrate in the sample. It is performed by using an Iodine solution. The test is known as the Iodine starch test. Starch test is used to check whether photosynthesis has occurred or not.
Why do we perform iodine test?
Answer : We perform iodine test to show the presence of starch. The presence of starch will be indicated by blue-black colour when iodine solution is poured on the surface of the leaf. If starch will be absent it will be indicated by brown colour.
Why do polysaccharides give iodine test?
Why is it an essential laboratory procedure? An iodine test can be used for the detection of starch in a given sample. The iodine test can help to distinguish starch from monosaccharides, disaccharides, and other polysaccharides. The iodine test is used for distinguishing between starch, glycogen, and carbohydrates.
How do you test for starch in a leaf?
- heat a plant leaf in boiling water for 30 seconds (this stops its chemical reactions)
- heat it in boiling ethanol for a few minutes (this removes most of its colour)
- wash with water and spread onto a white tile.
- add iodine solution from a dropping pipette.
How would you test the presence of starch in leaves in Short answer?
Presence of starch in leaves can be tested by iodine test. Iodine turns starch solution into blue-black colour. Pour few drops of dilute iodine solution on the boiled leaf. The leaf becomes blue-black which proves the presence of starch in it.
How would you test for starch in a plant leaf?
- Iodine solution is used to test leaves for the presence of starch. …
- After a few minutes, the parts of the leaf that contain starch turn the iodine from brown to blue/black.
- The leaf on the left is a variegated leaf.
Why does a test tube containing starch and saliva shows negative result for iodine test?
Saliva converts starch to sugars, which do not give blue-black colour with iodine.
What happens when iodine is added to boiled rice?
When boiled rice are added to the iodine solution then blue black colour appears because of presence of starch. when the boiled and chewed rice are added to the iodine solution then the colour of iodine will not change because of digestion of starch into sugars.
Why does the boiled rice after chewing not show the starch test?
Conclusion: Since test tube B has boiled rice which were chewed for 3-5 minutes, the saliva present in mouth has acted upon starch to break down into sugars. So it does not show any colour in presence of iodine solution.
How do you test for iodine in salt?
In the test, hydrogen peroxide reacts with iodide ion present in the salt sample (eq 3). Starch is also added to the testing mixture, so that any iodine produced will form a blue starch– iodine complex.
How do you test for iodine in salt at home?
To do this, pour one half cup of distilled water into a disposable cup, add one half teaspoon (tsp.) of laundry starch solution and then add five drops of the iodine antiseptic solution. Be careful when handling the iodine because it can stain. Stir well with a disposable plastic spoon.
What blood test checks iodine levels?
Thyroglobulin blood testing in adults can be used as a population measure of adequate iodine nutrition. Iodine is a common micronutrient from the diet.
How can iodine test be a good test in testing for saturated fat levels in cooking oil?
Principle of the test The most important application of the iodine value is to determine the amount of unsaturation contained in fatty acids. This unsaturation is in the form of double bonds which react with iodine compounds. The higher the iodine value, the more unsaturated fatty acid bonds are present in a fat.
Why do we check iodine value in oil?
Iodine numbers are often used to determine the amount of unsaturation in fats, oils and waxes. In fatty acids, unsaturation occurs mainly as double bonds which are very reactive towards halogens, the iodine in this case. Thus, the higher the iodine value, the more unsaturations are present in the fat.
What is the main purpose of huble's test?
(e) Huble’s Test: This test is used to know the degree of unsaturation in the given sample. Oils on reaction with Huble’s reagent fads the violet colour of iodine then it is unsaturated and if the colour persists then the given fat or oil is saturated.
What is the importance of acrolein test?
Acrolein test is used to detect the presence of glycerol or fat. When fat is treated strongly in the presence of a dehydrating agent like potassium bisulphate (KHSO4), the glycerol portion of the molecule is dehydrated to form an unsaturated aldehyde, acrolein that has a pungent irritating odour.