Does Net diffusion come to an end
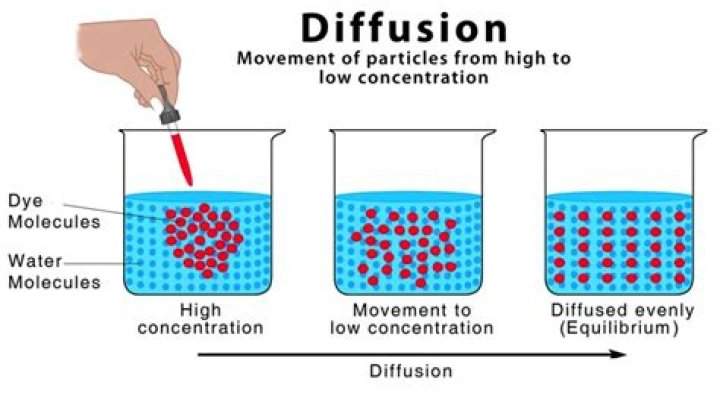
it is the random motion of the molecules that causes them to move from an area of high concentration to an area with a lower concentration. Diffusion will continue until the concentration gradient has been eliminated. … At equilibrium, movement of molecules does not stop.
At what point does net diffusion end?
The bigger the difference, the steeper the concentration gradient and the faster the molecules of a substance will diffuse. The direction of diffusion is said to be ‘down’ or ‘with’ the concentration gradient. Diffusion stops when the concentration of the substance is equal in both areas.
How does net diffusion work?
Net diffusion is when the flow of solute particle moving in one direction is bigger than the flow of solute particles moving in the opposite direction.
What is the end result of diffusion called?
Since diffusion moves materials from an area of higher concentration to the lower, it is described as moving solutes “down the concentration gradient.” The end result of diffusion is an equal concentration, or equilibrium, of molecules on both sides of the membrane. …What does net mean in diffusion?
Net Diffusion is a net movement (where the maximum particles moved) movement of particles from a region of higher concentration to lower concentration down the concentration gradient.
How does diffusion occur?
Diffusion occurs when particles spread. They move from a region where they are in high concentration to a region where they are in low concentration. Diffusion happens when the particles are free to move. This is true in gases and for particles dissolved in solutions – but diffusion does not occur in solids.
What does net movement mean?
The number of the molecules moving in the direction of the force minus the number of the molecules moving in other directions is the net movement of the molecules.
Does the net movement of molecules slow down as equilibrium is reached?
At equilibrium, movement of molecules does not stop. At equilibrium, there is equal movement of materials in both directions.What does no net movement mean?
This movement is known as a net movement where the molecules move in a particular direction according to their concentration. … The movement occurring is random and does not follow the concentration gradient. This stage is known as the no net movement stage, as molecules are moving randomly.
How long does diffusion last?Restricted diffusion typically occurs within 30-120 minutes after a cerebral infarction, returning to normal by 10-14 days. The principal mechanisms are thought to be: Increase in intracellular water.
Article first time published onHow does dissolving rely on diffusion?
Diffusion is the passage of particles from an area of higher concentration to an area of lower concentration. Dissolving is the bombardment of a solvent (e.g. water) on solute molecules (something that can dissolve, like sugar) forcing them apart. The solute mixes with a solvent to form a solution.
Does temperature affect net diffusion?
Temperature: Higher temperatures increase the energy and therefore the movement of the molecules, increasing the rate of diffusion.
How does heat affect the rate of diffusion?
The higher the temperature, the more kinetic energy the particles will have, so they will move and mix more quickly. The greater the surface area, the faster the rate of diffusion.
Does Net diffusion stop at equilibrium?
it is the random motion of the molecules that causes them to move from an area of high concentration to an area with a lower concentration. Diffusion will continue until the concentration gradient has been eliminated. … At equilibrium, movement of molecules does not stop.
What is net diffusion rate?
The rate of diffusion, dn/dt, is the change in the number of diffusing molecules inside the cell over time. Since the net movement of diffusing molecules depends on the concentration gradient, the rate of diffusion is directly proportional to the concentration gradient (dC/dx) across the membrane.
How do you describe the net movement of molecules?
Diffusion is defined as the net movement of molecules from an area of greater concentration to an area of lesser concentration. The molecules in a gas, a liquid or a solid are in constant motion due to their kinetic energy. Molecules are in constant movement and collide with each other.
How is tonicity affected by solutes?
Tonicity describes how an extracellular solution can change the volume of a cell by affecting osmosis. … A solution with low osmolarity has a greater number of water molecules relative to the number of solute particles; a solution with high osmolarity has fewer water molecules with respect to solute particles.
Why is net movement important in osmosis?
The net movement of water from a region of high water concentration to a region of low water concentration through a selectively permeable membrane. … This is because the selectively permeable membrane lets water molecules pass through much more rapidly than it lets sugar molecules pass through.
What is the direction of net movement?
The net movement of water (osmosis) is in the direction of increased solute concentrations. An easy way to visualize this rule is simply that the net water movement is from an area of high water concentration (little dissolved solute) to an area of low water concentration (high levels of solute). Animal cells.
Where does simple diffusion occur?
Simple passive diffusion occurs when small molecules pass through the lipid bilayer of a cell membrane. Facilitated diffusion depends on carrier proteins imbedded in the membrane to allow specific substances to pass through, that might not be able to diffuse through the cell membrane.
Where does diffusion take place?
Notably, diffusion takes place when a new chemical enters a cell’s cytosol, the fluid part of a cell’s insides. After new molecules enter a cell, they diffuse evenly through the cytosol. Note that they don’t enter the cell through diffusion; again, that’s osmosis.
Is diffusion active or passive?
Simple diffusion and osmosis are both forms of passive transport and require none of the cell’s ATP energy.
What is the difference between tonicity and osmotic pressure?
Osmotic pressure is the pressure of a solution against a semipermeable membrane to prevent water from flowing inward across the membrane. Tonicity is the measure of this pressure. … Usually, there is a higher concentration of solutes on one side of the membrane than the other.
How do you compare tonicity?
Tonicity is the concentration of a solution as compared to another solution. Concentration describes the amount of solutes dissolved by a solution. If a solution has a higher concentration of solutes (less water) than another it is said to be hypertonic.
When diffusion has reached equilibrium do molecules stop moving?
At equilibrium, movement of molecules does not stop. At equilibrium, there is equal movement of materials in both directions. If a molecule can pass freely through a cell membrane, it will cross the membrane by diffusion (Figure below).
When diffusion has reached equilibrium we say that the net movement of the molecules is zero Does this mean these molecules below would stop moving explain?
The molecules will not stop moving. The molecules are continuously moving even when they have reached equilibrium. It is just that the overall movement has moved from an area of high concentration to an area of low concentration.
How do molecules move during diffusion?
During diffusion, substances move from an area of high concentration to an area of low concentration, until the concentration becomes equal throughout a space. … There is net movement of molecules from the outside to the inside of the cell until the concentrations are equal on both sides.
Do molecules stop moving when diffusion stops?
Molecules are always moving. … When a molecule is diffusing, the molecule moves and when it reaches equilibrium, it means that the movement of the molecule from the higher concentration region to lower concentration stops but the molecules are always in a random movement about their fixed position.
What is meant by term diffusion?
diffusion, process resulting from random motion of molecules by which there is a net flow of matter from a region of high concentration to a region of low concentration.
Does diffusion move water?
Large quantities of water molecules constantly move across cell membranes by simple diffusion, often facilitated by movement through membrane proteins, including aquaporins.
Is dissolution a form of diffusion?
In this case, dissolution becomes a pure diffusional phenomenon. When a particle dissolves by pure diffusion, the concentration at every point away from the solid–liquid interface rises, but the concentration gradient at any distance from the particle decreases with time.